Efficacy of Low-Dose Fenfluramine on Adult Patients With Lennox-Gastaut Syndrome
- PMID: 40755616
- PMCID: PMC12317795
- DOI: 10.7759/cureus.87250
Efficacy of Low-Dose Fenfluramine on Adult Patients With Lennox-Gastaut Syndrome
Abstract
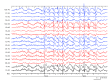
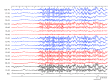
Purpose Fenfluramine has received approval for treating seizures in individuals with Lennox-Gastaut syndrome (LGS). Nonetheless, Phase III trials mainly focused on children, leaving limited information regarding its effectiveness and safety in adults. This case series aimed to elucidate these factors in adult patients. Methods This case series examined the clinical progression of adult patients diagnosed with LGS who received fenfluramine at our clinic following its approval in Japan in March 2024, extending through February 2025. Fenfluramine was administered to those experiencing drop attacks and generalized slow spike-and-wave complexes, fulfilling the conventional diagnostic criteria for LGS, and their responses to treatment were analyzed. Results This case series involved nine adult patients aged 18-49. All patients demonstrated efficacy, with seven (78%) experiencing at least a 50% reduction in seizure frequency. No one needed maintenance doses above the recommended starting minimum of 0.2 mg/kg/day, and five patients (56%) achieved seizure control with even lower doses. Remarkably, three patients who did not meet the latest 2022 diagnostic criteria also demonstrated positive responses. Adverse events were reported in five patients (56%), primarily including somnolence, diarrhea, and anorexia. In these situations, a dose reduction to half was necessary, yet efficacy was maintained. Conclusion Low-dose fenfluramine may effectively manage seizures in adult patients with LGS while reducing adverse effects. This case series provides a growing body of real-world data regarding the use of fenfluramine in this patient population.
Keywords: adult; fenfluramine; lennox-gastaut syndrome; low-dose; sigma-1.
Copyright © 2025, Takeda et al.
Conflict of interest statement
Human subjects: Informed consent for treatment and open access publication was obtained or waived by all participants in this study. The Institutional Review Ethics Board at Yokohama City University issued approval General 2024-047. Animal subjects: All authors have confirmed that this study did not involve animal subjects or tissue. Conflicts of interest: In compliance with the ICMJE uniform disclosure form, all authors declare the following: Payment/services info: All authors have declared that no financial support was received from any organization for the submitted work. Financial relationships: All authors have declared that they have no financial relationships at present or within the previous three years with any organizations that might have an interest in the submitted work. Other relationships: All authors have declared that there are no other relationships or activities that could appear to have influenced the submitted work.
Figures
Similar articles
-
Levetiracetam add-on for drug-resistant focal epilepsy: an updated Cochrane Review.Cochrane Database Syst Rev. 2012 Sep 12;2012(9):CD001901. doi: 10.1002/14651858.CD001901.pub2. Cochrane Database Syst Rev. 2012. PMID: 22972056 Free PMC article.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
Treatments for seizures in catamenial (menstrual-related) epilepsy.Cochrane Database Syst Rev. 2021 Sep 16;9(9):CD013225. doi: 10.1002/14651858.CD013225.pub3. Cochrane Database Syst Rev. 2021. PMID: 34528245 Free PMC article.
-
Rufinamide add-on therapy for refractory epilepsy.Cochrane Database Syst Rev. 2018 Apr 25;4(4):CD011772. doi: 10.1002/14651858.CD011772.pub2. Cochrane Database Syst Rev. 2018. Update in: Cochrane Database Syst Rev. 2020 Nov 8;11:CD011772. doi: 10.1002/14651858.CD011772.pub3. PMID: 29691835 Free PMC article. Updated.
-
Comparative efficacy and safety of stiripentol, cannabidiol and fenfluramine as first-line add-on therapies for seizures in Dravet syndrome: A network meta-analysis.Epilepsia Open. 2024 Apr;9(2):689-703. doi: 10.1002/epi4.12923. Epub 2024 Mar 1. Epilepsia Open. 2024. PMID: 38427284 Free PMC article.
References
-
- International League Against Epilepsy classification and definition of epilepsy syndromes with onset in childhood: position paper by the ILAE Task Force on Nosology and Definitions. Specchio N, Wirrell EC, Scheffer IE, et al. Epilepsia. 2022;63:1398–1442. - PubMed
-
- Long-term seizure outcome in 74 patients with Lennox-Gastaut syndrome: effects of incorporating MRI head imaging in defining the cryptogenic subgroup. Goldsmith IL, Zupanc ML, Buchhalter JR. Epilepsia. 2000;41:395–399. - PubMed
-
- Lennox-Gastaut syndrome: a comprehensive review. Asadi-Pooya AA. Neurol Sci. 2018;39:403–414. - PubMed
-
- Definition and natural history of Lennox-Gastaut syndrome. Camfield PR. Epilepsia. 2011;52 Suppl 5:3–9. - PubMed
-
- Lennox-Gastaut syndrome (childhood epileptic encephalopathy) Markand ON. J Clin Neurophysiol. 2003;20:426–441. - PubMed
LinkOut - more resources
Full Text Sources