Core-shell nanofibers for localized melanoma therapy delivering Pioglitazone nanoemulsions and gemcitabine dual loaded system
- PMID: 40760166
- PMCID: PMC12322006
- DOI: 10.1038/s41598-025-14483-1
Core-shell nanofibers for localized melanoma therapy delivering Pioglitazone nanoemulsions and gemcitabine dual loaded system
Abstract
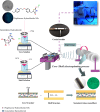
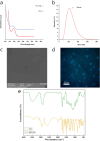
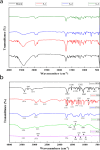
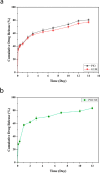
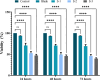
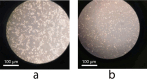
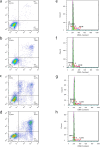
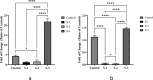
Melanoma is the most aggressive type of skin cancer and has very high rates of mortality. The primary objective of this study was to fabricate core-shell nanofibers as a drug delivery system using the coaxial electrospinning technique, which provides some distinct features. Polycaprolactone (PCL)-chitosan (CS)/polyvinyl alcohol (PVA) core-shell nanofibers embedded with Pioglitazone hydrochloride-loaded Nanoemulsions (PIO NEs), a general anti-diabetic drug, and Gemcitabine hydrochloride (GEM), a chemotherapeutic agent, were prepared to investigate the effects of combination of GEM and PIO against A375 melanoma skin cancer cells in vitro. The prepared PCL-CS/PVA-PIO NEs-GEM core-shell nanofibers exhibited sustained and controlled release profiles of GEM and PIO NEs over 14 days, which was fitted into various kinetic models. The data demonstrated the efficacy of nanoemulsions in improving the solubility and release of the poorly aqueous soluble drug PIO. The maximum amount released from the core-shell nanofibers reached 76.99 ± 1.5% of the GEM and 80.47 ± 2.01% of the PIO in a medium of pH 7.4. The nanofibers' morphology, chemical composition, weight loss, and swelling behavior were evaluated. MTT and flow cytometry analyses demonstrated that the combination of PIO and GEM effectively inhibited the growth of melanoma cancer cell lines by inhibiting proliferation with cell viability of 47.07 ± 2.5%, 45.36 ± 2.8%, and 39.79 ± 1.8% after 24, 48 and 72 h, inducing G0 /G1 phase arrest and apoptosis, and exhibited an enhanced combinatorial effect in A375 cells in vitro. Additionally, real-time PCR analysis confirmed the induction of apoptosis by measuring gene expression levels, suggesting that the mechanism is associated with the P53 and PPARγ pathways. The generated core-shell nanofibers exhibit properties that suggest their potential as an innovative local drug delivery system, suitable for direct implantation at the tumor site for melanoma treatment through a unique combination therapy of PIO and GEM.
Keywords: Chitosan; Core-shell nanofibers; Gemcitabine hydrochloride; Melanoma; Nanoemulsions; PCL; PVA; Pioglitazone hydrochloride.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests. Ethical approval: The authors have fully observed the ethical points in conducting the research and writing the results. All methods were carried out in accordance with relevant guidelines and regulations. All experimental protocols were approved by the Islamic Azad University Research Committee. Informed consent was obtained from all subjects and/or their legal guardian(s).
Figures


Similar articles
-
Selenium nanoparticles and paclitaxel co-delivery by a PCL based nanofibrous scaffold to enhance melanoma therapy.J Biomater Appl. 2025 Aug;40(2):165-180. doi: 10.1177/08853282251330724. Epub 2025 Mar 28. J Biomater Appl. 2025. PMID: 40152195
-
Preparation of poly(vinyl alcohol) nanofibers containing disulfiram-copper complex by electrospinning: a potential delivery system against melanoma.Daru. 2024 Dec;32(2):573-583. doi: 10.1007/s40199-024-00527-w. Epub 2024 Jul 4. Daru. 2024. PMID: 38963538 Free PMC article.
-
A rapid and systematic review of the clinical effectiveness and cost-effectiveness of paclitaxel, docetaxel, gemcitabine and vinorelbine in non-small-cell lung cancer.Health Technol Assess. 2001;5(32):1-195. doi: 10.3310/hta5320. Health Technol Assess. 2001. PMID: 12065068
-
Encapsulation of Gemcitabine on Porphyrin Aluminum Metal-Organic Framework by Mechano-Chemistry, Delayed Drug Release and Cytotoxicity to Pancreatic Cancer PANC-1 Cells.Molecules. 2024 Jul 4;29(13):3189. doi: 10.3390/molecules29133189. Molecules. 2024. PMID: 38999141 Free PMC article.
-
Systemic treatments for metastatic cutaneous melanoma.Cochrane Database Syst Rev. 2018 Feb 6;2(2):CD011123. doi: 10.1002/14651858.CD011123.pub2. Cochrane Database Syst Rev. 2018. PMID: 29405038 Free PMC article.
References
-
- Martins, N. et al. Advancements Melanoma Cancer Metastasis Models Pigment Cell. Melanoma Res., 36(2): 206–223. (2023). - PubMed
-
- Janani, I. et al. Selectivity and sensitivity of molybdenum oxide-polycaprolactone nanofiber composites on skin cancer: preliminary in-vitro and in-vivo implications. J. Trace Elem. Med Biol.49, 60–71 (2018). - PubMed
-
- Sajadimajd, S. et al. Advances on natural polyphenols as anticancer agents for skin cancer. Pharmacol. Res.151, 104584 (2020). - PubMed
-
- Simoes, M. F., Sousa, J. S. & Pais, A. C. Skin cancer and new treatment perspectives: A review. Cancer Lett.357 (1), 8–42 (2015). - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

