Evaluation of pre-treatment F-18 FDG PET/CT according to Mandard classification in locally advanced rectal cancer patients undergoing neoadjuvant chemoradiotherapy
- PMID: 40760425
- PMCID: PMC12323121
- DOI: 10.1186/s12885-025-14659-y
Evaluation of pre-treatment F-18 FDG PET/CT according to Mandard classification in locally advanced rectal cancer patients undergoing neoadjuvant chemoradiotherapy
Abstract
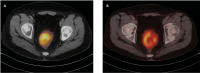
Background: This study primarily aimed to assess whether baseline [(18)F] fluorodeoxyglucose (FDG) PET/CT metabolic parameters-including SUVmax, metabolic tumor volume (MTV), and total lesion glycolysis (TLG)-can predict tumor regression grade (TRG) and survival in patients with locally advanced rectal cancer (LARC) undergoing neoadjuvant chemoradiotherapy (nCRT). In addition, secondary analyses were performed to identify other clinical and pathological variables associated with treatment response and prognosis.
Methods: This retrospective study included patients diagnosed with LARC who underwent nCRT followed by surgical resection between 2014 and 2023. Pre-treatment staging with F-18 FDG PET/CT was performed for all patients. Postoperative pathological response was assessed using the TRG system. Patients were categorized into two groups: complete responders (TRG1) and incomplete responders (TRG2-5). Demographic characteristics, PET/CT metabolic parameters (SUVmax, MTV, TLG), Carcinoembryonic antigen (CEA), Carbohydrate antigen 19 - 9 (CA19-9), and histopathological features (perforation, lymphovascular invasion [LVI], and perineural invasion [PNI]) were compared between the groups. Statistical analyses included chi-square tests, Mann-Whitney U tests, logistic regression models with odds ratios (ORs), and Kaplan-Meier survival analysis.
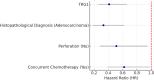
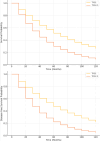
Results: A total of 151 patients were included. A statistically significant difference was found between TRG1 and TRG2-5 groups regarding family history (p = 0.034), CEA at diagnosis (p = 0.002), Ca19.9 after radiotherapy (p = 0.045), and presence of concurrent chemotherapy (CC) (p = 0.004). Significant differences were also observed in postoperative pathological features, including perforation (p = 0.045), LVI (p = 0.023), PNI (p = 0.031), and post-operative CEA levels (p = 0.001). In terms of outcomes, TRG1 was associated with better survival (p = 0.001), longer disease-free survival (p = 0.001), and overall survival (p = 0.001). Logistic regression identified independent predictors of complete response (TRG1): family history (OR: 5.08, p = 0.027), post-RT CEA (OR: 0.61, p = 0.015), post-op CEA (OR: 1.13, p = 0.012), perforation (OR: 20.93, p = 0.033), LVI (OR: 0.33, p = 0.042), and PNI (OR: 0.49, p = 0.045). Kaplan-Meier analysis demonstrated significantly longer overall survival in patients with TRG1 compared to TRG2-5 (log-rank p = 0.001). Similarly, disease-free survival was significantly better in the TRG1 group (log-rank p = 0.001). In the multivariate Cox regression model, TRG1 (HR: 0.41; 95% CI: 0.26-0.66; p = 0.001), CC (HR: 0.62; 95% CI: 0.39-0.98; p = 0.039), and absence of perforation (HR: 0.51; 95% CI: 0.28-0.95; p = 0.034) were found to be independent predictors of improved survival.
Conclusions: Baseline F-18 FDG PET/CT parameters, including SUVmax, MTV, and TLG, were not predictive of TRG or survival in patients with LARC undergoing neoadjuvant chemoradiotherapy. However, clinical and pathological variables such as family history, post-treatment CEA levels, perforation, lymphovascular invasion, and perineural invasion were significantly associated with TRG1. TRG1, histopathological subtype, perforation, and CC were also found to be independent predictors of survival. These findings suggest that while FDG-PET/CT may have limited utility in predicting treatment response, certain clinical and pathological features remain critical for outcome assessment.
Keywords: (18F) FDG PET/CT; Locally advanced rectal cancer; Mandard classification; Neoadjuvant chemoradiotherapy.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: This study was approved by the decision of University Faculty of Medicine Health Sciences Ethics Committee (Date: 23.11.2022 and approval no: 1582. Informed consent to participate was obtained from all participants in the study. The Declaration of Helsinki protocol was followed in the research protocol. Our study is a retrospective study. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures
Similar articles
-
Prognostic utility of serial 18F-FDG-PET/CT in patients with locally advanced rectal cancer who underwent tri-modality treatment.Br J Radiol. 2020 Jan;93(1105):20190455. doi: 10.1259/bjr.20190455. Epub 2019 Oct 23. Br J Radiol. 2020. PMID: 31617737 Free PMC article.
-
Early prediction of histopathological response of locally advanced rectal cancer after 1 week of preoperative radiochemotherapy using 18FDG PET-CT imaging: a prospective clinical validation study.Radiat Oncol. 2025 Aug 1;20(1):121. doi: 10.1186/s13014-025-02703-x. Radiat Oncol. 2025. PMID: 40751160 Free PMC article.
-
Does baseline [18F] FDG-PET/CT correlate with tumor staging, response after neoadjuvant chemoradiotherapy, and prognosis in patients with rectal cancer?Radiat Oncol. 2018 Oct 25;13(1):211. doi: 10.1186/s13014-018-1154-3. Radiat Oncol. 2018. PMID: 30359275 Free PMC article.
-
Potential Predictive Immune and Metabolic Biomarkers of Tumor Microenvironment Regarding Pathological and Clinical Response in Esophageal Cancer After Neoadjuvant Chemoradiotherapy: A Systematic Review.Ann Surg Oncol. 2024 Jan;31(1):433-451. doi: 10.1245/s10434-023-14352-z. Epub 2023 Sep 30. Ann Surg Oncol. 2024. PMID: 37777688 Free PMC article.
-
Hysterectomy with radiotherapy or chemotherapy or both for women with locally advanced cervical cancer.Cochrane Database Syst Rev. 2015 Apr 7;(4):CD010260. doi: 10.1002/14651858.CD010260.pub2. Cochrane Database Syst Rev. 2015. Update in: Cochrane Database Syst Rev. 2022 Aug 22;8:CD010260. doi: 10.1002/14651858.CD010260.pub3. PMID: 25847525 Updated.
References
-
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–49. 10.3322/caac.21660. - PubMed
-
- Keller DS, Berho M, Perez RO, Wexner SD, Chand M. The multidisciplinary management of rectal cancer. Nat Rev Gastroenterol Hepatol. 2020;17(7):414–29. 10.1038/s41575-020-0275-y. - PubMed
-
- Costas-Chavarri A, Nandakumar G, Temin S, Lopes G, Cervantes A, Cruz Correa M, Engineer R, Hamashima C, Ho GF, Huitzil FD, Malekzadeh Moghani M, Sharara AI, Stern MC, Teh C, Vazquez Manjarrez SE, Verjee A, Yantiss R, Shah MA. Treatment of patients with early-stage colorectal cancer: ASCO resource-stratified guideline. J Glob Oncol. 2019;5:1–19. 10.1200/JGO.18.00214. - PMC - PubMed
-
- Aguiar Junior S, Oliveira MM, Silva D, Mello CAL, Calsavara VF, Curado MP. Survival of patients with colorectal cancer in a cancer center. Arq Gastroenterol. 2020;57(2):172–7. 10.1590/S0004-2803.202000000-32. - PubMed
-
- Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024. 10.3322/caac.21834. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

