Integrating Deep Learning and Real-Time Imaging to Visualize In Situ Self-Assembly of Self-Healing Interpenetrating Polymer Networks Formed by Protein and Polysaccharide Fibers
- PMID: 40762431
- PMCID: PMC12371690
- DOI: 10.1021/acsami.5c11459
Integrating Deep Learning and Real-Time Imaging to Visualize In Situ Self-Assembly of Self-Healing Interpenetrating Polymer Networks Formed by Protein and Polysaccharide Fibers
Abstract
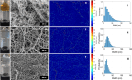
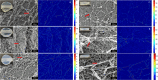
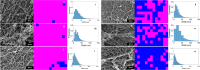
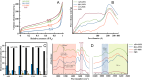
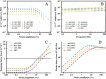
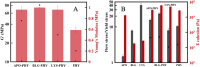
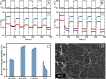
Fibrillar protein hydrogels are promising sustainable biomaterials for biomedical applications, but their practical use is often limited by insufficient mechanical strength and stability. To address these challenges, we transformed native proteins into amyloid fibrils (AFs) and incorporated a fibrillar polysaccharide, phytagel (PHY), to engineer interpenetrating polymer network (IPN) hydrogels. Notably, we report for the first time the formation of an amyloid-based hydrogel from apoferritin (APO), with PHY reinforcing the network's mechanical integrity. In situ self-assembly of APO within the PHY matrix yields fully natural, biopolymer-based IPNs. Rheological analyses confirm synergistic interactions between AF and PHY fibers, with the composite hydrogels exhibiting significantly enhanced viscoelastic moduli compared with individual components. The AF-PHY hydrogels also demonstrate excellent self-healing behavior, rapidly restoring their storage modulus after high-strain deformation. A major advancement of this study is the application of deep learning (DL)-based image analysis, using convolutional neural networks, to automate the identification, segmentation, and quantification of fibrillar components in high-resolution scanning electron microscopy images. This AI-driven method enables precise differentiation between AF and PHY fibers and reveals the three-dimensional microarchitecture of the IPN, overcoming key limitations of traditional image analysis. Complementary real-time confocal laser scanning microscopy, with selective fluorescent labeling of protein and polysaccharide components, further validates the IPN structure of the hybrid hydrogels. Our results demonstrate that DL significantly enhances structural characterization and provides insights into gelation processes. This approach sets a new guide for the analysis of complex soft materials and underlines the potential of AF-PHY hydrogels as mechanically robust, self-healing, and fully sustainable biomaterials for biomedical engineering applications.
Keywords: IPN networks; deep learning; fibrillar polysaccharide; hydrogels; protein fibers; real-time CLSM imaging.
Figures



Similar articles
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
Anterior Approach Total Ankle Arthroplasty with Patient-Specific Cut Guides.JBJS Essent Surg Tech. 2025 Aug 15;15(3):e23.00027. doi: 10.2106/JBJS.ST.23.00027. eCollection 2025 Jul-Sep. JBJS Essent Surg Tech. 2025. PMID: 40821726 Free PMC article.
-
Antibacterial collagen-guar gum hydrogels with zeolitic imidazolate framework-67 (ZIF-67): an innovative platform for advanced wound healing.J Biomater Sci Polym Ed. 2025 Sep;36(13):1827-1852. doi: 10.1080/09205063.2025.2486859. Epub 2025 Apr 1. J Biomater Sci Polym Ed. 2025. PMID: 40168277
-
Thermoresponsive polymers for cell support: poloxamers as a case study of promise and challenge.J Mater Chem B. 2025 Aug 6;13(31):9351-9376. doi: 10.1039/d5tb00588d. J Mater Chem B. 2025. PMID: 40662391 Review.
-
Management of urinary stones by experts in stone disease (ESD 2025).Arch Ital Urol Androl. 2025 Jun 30;97(2):14085. doi: 10.4081/aiua.2025.14085. Epub 2025 Jun 30. Arch Ital Urol Androl. 2025. PMID: 40583613 Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

