Tiantan vaccinia virus-based vaccine with promising safety provides sustained protection against mpox in non-human primates
- PMID: 40764511
- PMCID: PMC12325645
- DOI: 10.1038/s41467-025-62594-0
Tiantan vaccinia virus-based vaccine with promising safety provides sustained protection against mpox in non-human primates
Abstract
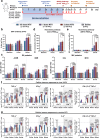
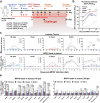
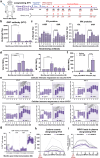
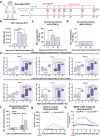
The World Health Organization (WHO) has declared the mpox outbreak a public health emergency of international concern (PHEIC). Safe and efficient vaccines against the mpox virus (MPXV) are urgently needed to impede the surge in cases. Here, we report the results of a preclinical study employing different dosing strategies on a vaccine candidate named NTV, obtained via targeted gene deletion in the Tiantan strain vaccinia virus, resulting in a replication-deficient variant. Following optimisation of the NTV immunization dose and confirmation of its protective efficacy against MPXV in a mouse model, we demonstrate that a two-shot NTV regimen in macaques elicits significant neutralizing antibody and cellular immune responses, providing efficient protection against MPXV challenge. Notably, we find that a single NTV dose or long-term immunization in macaques offer effective protection against moderate or severe mpox disease by enhancing cellular immunity and rapidly evoking neutralizing antibodies. These results demonstrate the vaccine's potential for emergency use and for long-lasting protection. Safety evaluations show no adverse effects in macaques receiving triple the standard dosage in three consecutive injections. These findings highlight the potential of the NTV vaccine candidate with key advantages, including robust immunogenicity, sustained protective efficacy, and safety in preclinical settings.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures


References
-
- Musambi, E. Mpox deaths rise by 107 in a week as Africa CDC calls the toll unacceptable. https://apnews.com/article/mpox-africa-cdc-outbreak-deaths-testing-vacci... (2024).
-
- Grabenstein, J. D. & Hacker, A. Vaccines against mpox: MVA-BN and LC16m8. Expert Rev. Vaccines23, 796–811 (2024). - PubMed
-
- WHO. Mpox global strategic preparedness and response plan. https://www.who.int/publications/m/item/mpox-global-strategic-preparedne... (2024). - PMC - PubMed
-
- WHO. Strategic framework for enhancing prev ention and control of mpox-2024-2027. https://www.who.int/publications/i/item/9789240092907 (2024).
-
- Organization, W. H. Smallpox and mpox (orthopoxviruses): WHO Position Paper. https://www.who.int/publications/i/item/who-wer-9934-429-456 (2024).
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

