This is a preprint.
Concerted actions of distinct serotonin neurons orchestrate female pup care behavior
- PMID: 40766669
- PMCID: PMC12324472
- DOI: 10.1101/2025.07.31.667987
Concerted actions of distinct serotonin neurons orchestrate female pup care behavior
Abstract
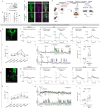
In many mammalian species, female behavior towards infant conspecifics changes across reproductive stages. Sexually naïve females interact minimally or aggressively with infants, whereas the same animals exhibit extensive care behavior, even towards unrelated infants, after parturition1-6. Here, we discovered that two distinct sets of serotonin neurons collectively mediate this dramatic transition in maternal behavior-serotonin neurons projecting to the medial preoptic area (mPOA) promote pup care in mothers, whereas those projecting to the bed nucleus of the stria terminalis (BNST) suppress pup interaction in virgin female mice. Disrupting serotonin synthesis in either of these subpopulations or stimulating either subpopulation is sufficient to toggle pup-directed behavior between that displayed by virgin females and that of lactating mothers. In virgin female mice, the first pup interaction triggers an increase in serotonin release in BNST but a decrease in mPOA. In mothers, serotonin activity becomes greatly elevated in mPOA during pup interactions. Acute interruption of serotonin signaling locally in either mPOA or BNST disrupts the stage-dependent switch in pup care. Together, these results highlight how functionally distinct serotonin subpopulations orchestrate social behaviors appropriate for a given reproductive state, and suggest a circuit logic for how a neuromodulator coordinates adaptive behavioral changes across life stages.
Conflict of interest statement
Competing interests: The authors declare that they have no competing interests.
Figures











Similar articles
-
Concerted actions of distinct serotonin neurons orchestrate female pup care behavior.Res Sq [Preprint]. 2025 Aug 4:rs.3.rs-7134286. doi: 10.21203/rs.3.rs-7134286/v1. Res Sq. 2025. PMID: 40799751 Free PMC article. Preprint.
-
Prepronociceptin-Expressing Neurons in the Bed Nucleus of the Stria Terminalis Signal Escape Behavior.Biol Psychiatry Glob Open Sci. 2025 May 22;5(5):100538. doi: 10.1016/j.bpsgos.2025.100538. eCollection 2025 Sep. Biol Psychiatry Glob Open Sci. 2025. PMID: 40678693 Free PMC article.
-
Antiretrovirals for reducing the risk of mother-to-child transmission of HIV infection.Cochrane Database Syst Rev. 2011 Jul 6;(7):CD003510. doi: 10.1002/14651858.CD003510.pub3. Cochrane Database Syst Rev. 2011. PMID: 21735394
-
Antiretrovirals for reducing the risk of mother-to-child transmission of HIV infection.Cochrane Database Syst Rev. 2007 Jan 24;(1):CD003510. doi: 10.1002/14651858.CD003510.pub2. Cochrane Database Syst Rev. 2007. Update in: Cochrane Database Syst Rev. 2011 Jul 06;(7):CD003510. doi: 10.1002/14651858.CD003510.pub3. PMID: 17253490 Updated.
-
Sexual Harassment and Prevention Training.2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 36508513 Free Books & Documents.
References
-
- McCarthy M. M. & Vom Saal F. S. The influence of reproductive state on infanticide by wild female house mice (Mus musculus). Physiol. Behav. 35, 843–849 (1985). - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
