Multimodal Inhibition of Pectobacterium brasiliense Virulence by the Citrus Flavanone Naringenin
- PMID: 40767039
- PMCID: PMC12371876
- DOI: 10.1021/acs.jafc.5c04312
Multimodal Inhibition of Pectobacterium brasiliense Virulence by the Citrus Flavanone Naringenin
Abstract
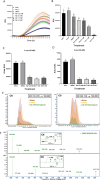
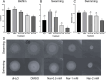
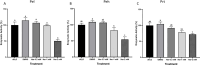
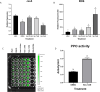
Naringenin, a flavanone from citrus, was studied for its ability to reduce virulence in Pectobacterium, a phytopathogen causing soft rot disease in crop plants. Naringenin downregulated quorum sensing (QS) and suppressed critical virulence determinants in Pectobacterium brasiliense Pb1692, including plant cell wall-degrading enzymes, bacterial motility, and biofilm formation, consequently reducing disease symptoms in two host plants. Molecular docking simulations revealed a plausible binding mode for naringenin within the QS protein ExpI, which were maintained during microsecond-long Molecular Dynamics simulations. These simulations provided atomic-scale insight into specific interactions and estimated binding free energies, supporting naringenin's QS inhibition mode of action. In contrast, S-adenosyl methionine, the natural ligand of ExpI, was unable to maintain a stable binding mode in the ExpI site during simulations. Beyond QS disruption, naringenin induced reactive oxygen species accumulation and compromised DNA repair, indicating a multimodal mechanism of action. Despite these promising findings, naringenin's limited aqueous solubility challenges practical applications.
Keywords: Pectobacterium brasiliense; ROS; molecular docking; molecular dynamics; naringenin; quorum sensing inhibitor.
Figures




Similar articles
-
Plant phenolic volatiles inhibit quorum sensing in pectobacteria and reduce their virulence by potential binding to ExpI and ExpR proteins.Sci Rep. 2016 Dec 1;6:38126. doi: 10.1038/srep38126. Sci Rep. 2016. PMID: 27905512 Free PMC article.
-
Plant phenolic acids affect the virulence of Pectobacterium aroidearum and P. carotovorum ssp. brasiliense via quorum sensing regulation.Mol Plant Pathol. 2016 May;17(4):487-500. doi: 10.1111/mpp.12295. Epub 2015 Sep 7. Mol Plant Pathol. 2016. PMID: 26177258 Free PMC article.
-
Multifaceted quorum-sensing inhibiting activity of 3-(Benzo[d][1,3]dioxol-4-yl)oxazolidin-2-one mitigates Pseudomonas aeruginosa virulence.Virulence. 2025 Dec;16(1):2479103. doi: 10.1080/21505594.2025.2479103. Epub 2025 Mar 19. Virulence. 2025. PMID: 40104940
-
FDA-approved drugs for targeting virulence of Pseudomonas aeruginosa: A drug repurposing approach to combat multidrug resistance.Microb Pathog. 2025 Sep;206:107781. doi: 10.1016/j.micpath.2025.107781. Epub 2025 Jun 3. Microb Pathog. 2025. PMID: 40473130 Review.
-
A critical review on innovative targets for signal disruption in Enterococcus faecalis infection management.Microb Pathog. 2025 Oct;207:107876. doi: 10.1016/j.micpath.2025.107876. Epub 2025 Jul 4. Microb Pathog. 2025. PMID: 40617451 Review.
References
-
- Gustafsson, J. ; Cederberg, C. ; Sonesson, U. ; Emanuelsson, A. . The Methodology of the FAO Study: Global Food Losses and Food Waste–Extent, Causes and Prevention–FAO, 2011; SIK Institutet för livsmedel och bioteknik, 2013.
-
- Ma B., Hibbing M. E., Kim H. S., Reedy R. M., Yedidia I., Breuer J., Breuer J., Glasner J. D., Perna N. T., Kelman A., Charkowski A. O.. Host range and molecular phylogenies of the soft rot enterobacterial genera pectobacterium and dickeya. Phytopathology. 2007;97(9):1150–63. doi: 10.1094/PHYTO-97-9-1150. - DOI - PubMed
-
- Tapia-Rodriguez M. R., Bernal-Mercado A. T., Gutierrez-Pacheco M. M., Vazquez-Armenta F. J., Hernandez-Mendoza A., Gonzalez-Aguilar G. A., Martinez-Tellez M. A., Nazzaro F., Ayala-Zavala J. F.. Virulence of Pseudomonas aeruginosa exposed to carvacrol: alterations of the Quorum sensing at enzymatic and gene levels. Journal of cell communication and signaling. 2019;13(4):531–537. doi: 10.1007/s12079-019-00516-8. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

