Phosphomannomutase 2-congenital disorder of glycosylation: exploring the role of N-glycosylation on the endocrine axes
- PMID: 40771275
- PMCID: PMC12325039
- DOI: 10.3389/fendo.2025.1594118
Phosphomannomutase 2-congenital disorder of glycosylation: exploring the role of N-glycosylation on the endocrine axes
Abstract
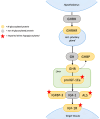
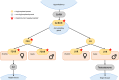
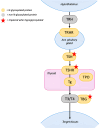
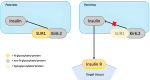
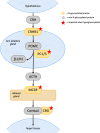
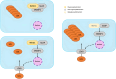
Congenital disorders of glycosylation (CDG) are a heterogeneous group of inborn errors of metabolism caused by impaired protein glycosylation. Among these, PMM2-CDG, caused by defective phosphomannomutase 2 activity and affecting protein N-glycosylation, is the most prevalent. As glycoproteins are involved in almost every physiological process, the clinical manifestations in PMM2-CDG are diverse and multisystemic. In the endocrine system, glycoproteins are present in every axis, acting as hormones, prohormones, receptors, enzymes, and transport proteins. Hypoglycosylation can alter hormonal function on multiple levels. As a result, endocrinopathies are frequently part of the clinical spectrum of PMM2-CDG, particularly hypergonadotrophic hypogonadism and pubertal abnormalities in female patients. Symptoms of endocrine involvement, especially hyperinsulinemic hypoglycemia and failure to thrive during infancy, can be the presenting sign of the disease. The clinical spectrum of PMM2-CDG endocrinopathy is variable; for example, thyroid involvement can range from isolated transitory hyperthyrotropinemia to clinical hypothyroidism. Some endocrine abnormalities, such as adrenal insufficiency, are uncommon and probably underdiagnosed in PMM2-CDG. The new insights into the role of N-glycosylation on the endocrine system over the past twenty years have deepened our understanding of this complex disorder and should enable us to improve and personalize the clinical management of these patients.
Keywords: N-glycosylation; congenital disorder of glycosylation; endocrine dysfunction; hypergonadotropic hypogonadism; hypoglycosylation.
Copyright © 2025 Del Medico, Ferri, Procopio, Annibalini, Barbieri, Barone, Guerrini, Morrone and Stagi.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
In Silico Analysis of Phosphomannomutase-2 Dimer Interface Stability and Heterodimerization with Phosphomannomutase-1.Molecules. 2025 Jun 15;30(12):2599. doi: 10.3390/molecules30122599. Molecules. 2025. PMID: 40572562 Free PMC article.
-
Multiorgan involvement and genetic spectrum of 20 Chinese patients with PMM2-CDG.Mol Genet Metab. 2025 Aug;145(4):109178. doi: 10.1016/j.ymgme.2025.109178. Epub 2025 Jun 15. Mol Genet Metab. 2025. PMID: 40555085
-
Frontiers in congenital disorders of glycosylation consortium, a cross-sectional study report at year 5 of 280 individuals in the natural history cohort.Mol Genet Metab. 2024 Aug;142(4):108509. doi: 10.1016/j.ymgme.2024.108509. Epub 2024 Jun 6. Mol Genet Metab. 2024. PMID: 38959600 Free PMC article.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
Liver involvement in congenital disorders of glycosylation (CDG). A systematic review of the literature.J Inherit Metab Dis. 2017 Mar;40(2):195-207. doi: 10.1007/s10545-016-0012-4. Epub 2017 Jan 20. J Inherit Metab Dis. 2017. PMID: 28108845
References
-
- Altassan R, Péanne R, Jaeken J, Barone R, Bidet M, Borgel D, et al. International clinical guidelines for the management of phosphomannomutase 2-congenital disorders of glycosylation: Diagnosis, treatment and follow up. J Inherit Metab Dis. (2019) 42:5–28. doi: 10.1002/JIMD.12024, PMID: - DOI - PubMed
-
- Muthusamy K, Perez-Ortiz JM, Ligezka AN, Altassan R, Johnsen C, Schultz MJ, et al. Neurological manifestations in PMM2-congenital disorders of glycosylation (PMM2-CDG): Insights into clinico-radiological characteristics, recommendations for follow-up, and future directions. Genet Med. (2024) 26. doi: 10.1016/j.gim.2023.101027, PMID: - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

