Mobiluncus mulieris alters the transcriptomic profile of cervicovaginal epithelial cells, shedding light on molecular drivers of adverse reproductive outcomes
- PMID: 40775239
- PMCID: PMC12331918
- DOI: 10.1038/s41522-025-00784-w
Mobiluncus mulieris alters the transcriptomic profile of cervicovaginal epithelial cells, shedding light on molecular drivers of adverse reproductive outcomes
Abstract
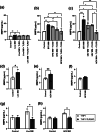
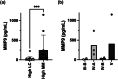
The vaginal microbiota is critical for reproductive health, and its disruption, particularly the loss of Lactobacillus spp. and dominance of anaerobes such as Mobiluncus mulieris (community state type IV, CST IV), is associated with bacterial vaginosis, sexually transmitted infections, and adverse reproductive outcomes, including preterm birth (PTB). While Gardnerella spp. have been widely studied, the role of M. mulieris remains poorly understood. This study used an unbiased discovery approach to examine host-microbe interactions driven by M. mulieris across distinct epithelial barriers of the lower reproductive tract. RNA sequencing revealed that live bacteria, cell-free supernatant, and bacterial extracellular vesicles (bEVs) each induced unique transcriptional responses in epithelial cells. All three components activated immune and inflammatory pathways, with bEVs eliciting the strongest response, particularly via toll-like receptor (TLR) 2 and TLR5 signaling. M. mulieris also altered extracellular matrix (ECM) remodeling pathways, including upregulation of matrix metalloproteinase 9 (MMP9), a key mediator linked to PTB. These findings were supported by clinical data showing elevated MMP9 in pregnant women with M. mulieris-containing vaginal microbiota. Collectively, these results highlight the broad impact of M. mulieris on epithelial responses and identify mechanisms by which specific anaerobes contribute to inflammation and ECM disruption in adverse reproductive outcomes.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: J.R. is co-founder of LUCA Biologics, a biotechnology company focusing on translating microbiome research into live biotherapeutic drugs for women’s health. All other authors declare that they have no competing interests.
Figures




References
-
- van de Wijgert, J. H. H. M. & Jespers, V. The global health impact of vaginal dysbiosis. Res. Microbiol.168, 859–864 (2017). - PubMed
MeSH terms
Substances
Grants and funding
- R01 HD102318/HD/NICHD NIH HHS/United States
- R01HD098867/National Institutes of Health (NIH) National Institute of Child Health and Human Development (NICHD)
- R01 NR014784/NR/NINR NIH HHS/United States
- R01 HD098867/HD/NICHD NIH HHS/United States
- R01NR014784/National Institute for Nursing Research of the National Institutes of Health
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

