Expansion of the antibacterial spectrum of symmetrical amino acid-paired antifungal peptides through structural optimization
- PMID: 40775273
- PMCID: PMC12333139
- DOI: 10.1186/s12866-025-04160-8
Expansion of the antibacterial spectrum of symmetrical amino acid-paired antifungal peptides through structural optimization
Abstract
Background: Fungal infections often co-occur with antibiotic-resistant bacterial infections, posing clinical treatment challenges. Antimicrobial peptides (AMPs) are considered promising therapeutic alternatives due to their low potential for inducing drug resistance. This study aimed to enhance the antibacterial potency of an existing antifungal peptide through optimization, developing a dual-function peptide targeting both fungal and bacterial pathogens.
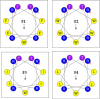
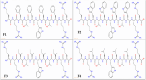
Methods: We designed peptides F1-F4 from the symmetrical amino acid-paired antifungal peptide P19 through threonine (T) substitution and hydrophobic moment (µHrel) adjustment. Then, we assessed their antifungal and antibacterial activities against reference and clinically isolated strains by minimum inhibitory concentrations (MICs), evaluated their toxicity to human red blood cells, and explored the membrane-associated mechanisms.
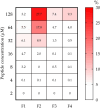
Results: Peptide F4 was the most promising candidate due to its potent antifungal and antibacterial activities, lack of inhibitory effect on beneficial lactobacilli at concentrations effective against pathogens, and low hemolytic activity. F4 also exhibited strong binding affinity to lipopolysaccharides (LPS) and induced bacterial membrane depolarization and permeabilization.
Conclusions: Our findings demonstrated that T substitution and hydrophobic moment adjustment effectively enhanced antibacterial activity of the antifungal peptide P19, making peptide F4 a strong candidate for both fungal and bacterial infections.
Keywords: Hydrophobicity; Probiotic; Resistant bacteria; Resistant fungi; Threonine.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Not applicable. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures






Similar articles
-
Broad-spectrum activity of membranolytic cationic macrocyclic peptides against multi-drug resistant bacteria and fungi.Eur J Pharm Sci. 2024 Jun 1;197:106776. doi: 10.1016/j.ejps.2024.106776. Epub 2024 Apr 23. Eur J Pharm Sci. 2024. PMID: 38663759 Free PMC article.
-
Enhancing the selectivity and conditional sensitivity of an antimicrobial peptide through cleavage simulations and homoarginine incorporation to combat drug-resistant bacteria.Sci Rep. 2025 Jul 1;15(1):21798. doi: 10.1038/s41598-025-06522-8. Sci Rep. 2025. PMID: 40594393 Free PMC article.
-
Optimized strategies for designing antimicrobial peptides targeting multidrug-resistant Gram-negative bacteria.Biomed Pharmacother. 2025 Aug;189:118264. doi: 10.1016/j.biopha.2025.118264. Epub 2025 Jun 19. Biomed Pharmacother. 2025. PMID: 40540884
-
AI-Driven Antimicrobial Peptide Discovery: Mining and Generation.Acc Chem Res. 2025 Jun 17;58(12):1831-1846. doi: 10.1021/acs.accounts.0c00594. Epub 2025 Jun 3. Acc Chem Res. 2025. PMID: 40459283 Free PMC article. Review.
-
Interventions to improve antibiotic prescribing practices for hospital inpatients.Cochrane Database Syst Rev. 2013 Apr 30;(4):CD003543. doi: 10.1002/14651858.CD003543.pub3. Cochrane Database Syst Rev. 2013. Update in: Cochrane Database Syst Rev. 2017 Feb 09;2:CD003543. doi: 10.1002/14651858.CD003543.pub4. PMID: 23633313 Updated.
References
-
- Hendrickson JA, Hu C, Aitken SL, Beyda N. Antifungal resistance: a concerning trend for the present and future. Curr Infect Dis Rep. 2019;21(12):47. - PubMed
-
- Schwartz IS, Patterson TF. The emerging threat of antifungal resistance in transplant infectious diseases. Curr Infect Dis Rep. 2018;20(3):2. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

