Beta cyclodextrin stabilized cupric oxide nanoparticles assisted thermal therapy for lung tumor and its effective in vitro anticancer activity
- PMID: 40775400
- PMCID: PMC12332176
- DOI: 10.1038/s41598-025-96578-3
Beta cyclodextrin stabilized cupric oxide nanoparticles assisted thermal therapy for lung tumor and its effective in vitro anticancer activity
Abstract
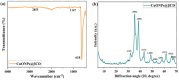
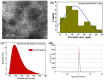
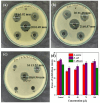
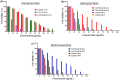
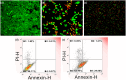
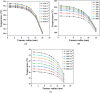
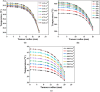
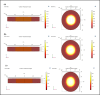
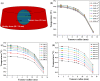
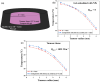
The unique physicochemical properties of cupric oxide nanoparticles (CuO NPs) make them suitable for a wide range of therapeutic applications. Here, we synthesized β-cyclodextrin (βCD) capped CuO NPs (CuONPs@βCD) using a simple reduction process. The formation and physicochemical characteristics were identified via different spectroscopic techniques. The CuONPs@βCD displayed antimicrobial activity as good as commercial drugs. Dimethyl thiazolyl tetrazolium bromide (MTT) assay was carried out to assess the anticancer properties of CuONPs@βCD against A549 lung cancer cells. The result demonstrated that the anticancer activity of CuONPs@βCD with IC50 values of 41.06 ± 0.05 and 19.46 µg/mL at 24 and 48-h incubation period, respectively. CuONPs@βCD exhibited anticancer activity on A549 lung cancer cells while having less adverse effects on normal cells. Annexin V-FITC/PI assay, reactive oxygen species (ROS) analysis, disruption of mitochondrial membrane potential (Δψm), and AO/EB apoptosis studies in A549 cells revealed significant apoptotic impact of CuONPs@βCD when compared to the control. Moreover, thermal therapy study of CuONPs@βCD in lung tumor using COMSOL Multiphysics has been reported. Our investigation revealed Case III, where the temperature distribution at the top surface of the tumor is best and may be the most effective way to treat lung cancer. It was found that an incident flux of 8000 Wm- 2 for 900 s and an extinction coefficient of 8.266 m- 1 for CuONPs@βCD were the best conditions for reaching a temperature of 43.63 °C across the whole tumor area. Thus, these findings open new research opportunities and potential use of CuONPs@βCD for biological applications.
Keywords: Anticancer; Antimicrobial; COMSOL multiphysics; CuONPs@βCD; Lung cancer; Temperature distribution.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests.
Figures




Similar articles
-
Investigation of antioxidant, antibacterial, anticancer and wound healing properties of eco-friendly synthesized copper oxide nanoparticles from Plumeria rubra leaf extract.Bioprocess Biosyst Eng. 2025 Aug;48(8):1295-1310. doi: 10.1007/s00449-025-03176-8. Epub 2025 May 21. Bioprocess Biosyst Eng. 2025. PMID: 40397157
-
Yttrium oxide nanoparticles induce selective cytotoxicity, genomic instability and ROS mitochondrial P53 mediated apoptosis in human pancreatic cancer cells.Sci Rep. 2025 Jun 20;15(1):20144. doi: 10.1038/s41598-025-05088-9. Sci Rep. 2025. PMID: 40542009 Free PMC article.
-
Influence of capping agents on physicochemical properties and leukemic cytotoxicity of copper oxide nanoparticles biosynthesized using Caesalpinia sappan extract.PLoS One. 2025 Jun 26;20(6):e0326791. doi: 10.1371/journal.pone.0326791. eCollection 2025. PLoS One. 2025. PMID: 40569954 Free PMC article.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
References
-
- Parayath, N. et al. Strategies for targeting cancer immunotherapy through modulation of the tumor microenvironment. Regen Eng. Transl Med.6, 29–49. 10.1007/s40883-019-00113-6 (2020).
-
- Datta, N. R. et al. Local hyperthermia combined with radiotherapy and-/or chemotherapy: Recent advances and promises for the future. Cancer Treat. Rev.41, 742–753. 10.1016/j.ctrv.2015.05.009 (2015). - PubMed
-
- Dou, J. P., Zhou, Q. F., Liang, P. & Yu, J. Advances in nanostructure-mediated hyperthermia in tumor therapies. Curr. Drug Metab.19, 85–93. 10.2174/1389200219666180129141757 (2018). - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

