From mitochondrial dysregulation to ferroptosis: Exploring new strategies and challenges in radioimmunotherapy (Review)
- PMID: 40776761
- PMCID: PMC12331309
- DOI: 10.3892/ijo.2025.5781
From mitochondrial dysregulation to ferroptosis: Exploring new strategies and challenges in radioimmunotherapy (Review)
Abstract
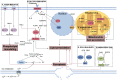
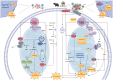
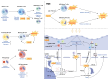
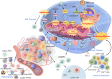
Ferroptosis is an iron‑dependent, lipid peroxidation‑driven form of regulated immunogenic cell death (ICD). ICD has demonstrated potential to overcome resistance to conventional cancer therapies, enhancing the efficacy of treatments such as chemotherapy, radiotherapy, immunotherapy and photodynamic therapy. Notably, in the context of radiotherapy, ferroptosis serves a key role, particularly when combined with radioimmunotherapy. Mitochondria are central to the regulation of radiation‑induced oxidative stress and the remodeling of the immune microenvironment, and they undergo characteristic morphological changes during the ferroptotic process. However, the precise regulatory association between mitochondrial dysfunction and ferroptosis remains incompletely understood, and there is an ongoing debate regarding this complex interaction. The present review aimed to explore the mechanisms through which mitochondria and ferroptosis interact in the context of radiotherapy, with a focus on how ferroptosis exacerbates mitochondrial dysfunction. Additionally, the present review proposed novel strategies leveraging radioimmunotherapy to offer more precise and effective approaches for cancer treatment.
Keywords: ferroptosis; mitochondrial dysregulation; radioimmunotherapy.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Similar articles
-
Radiotherapy elicits immunogenic cell death and metabolic shifts in the tumor microenvironment: implications for immunotherapy.Int J Med Sci. 2025 Jul 11;22(13):3277-3291. doi: 10.7150/ijms.109515. eCollection 2025. Int J Med Sci. 2025. PMID: 40765564 Free PMC article. Review.
-
Short-Term Memory Impairment.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31424720 Free Books & Documents.
-
Ferroptosis: a key driver and therapeutic target in the pathogenesis of acute respiratory distress syndrome.Front Immunol. 2025 Jul 22;16:1567980. doi: 10.3389/fimmu.2025.1567980. eCollection 2025. Front Immunol. 2025. PMID: 40766305 Free PMC article. Review.
-
The Mutual Regulatory Role of Ferroptosis and Immunotherapy in Anti-tumor Therapy.Apoptosis. 2024 Oct;29(9-10):1291-1308. doi: 10.1007/s10495-024-01988-9. Epub 2024 Jun 9. Apoptosis. 2024. PMID: 38853203 Free PMC article. Review.
-
Systemic treatments for metastatic cutaneous melanoma.Cochrane Database Syst Rev. 2018 Feb 6;2(2):CD011123. doi: 10.1002/14651858.CD011123.pub2. Cochrane Database Syst Rev. 2018. PMID: 29405038 Free PMC article.
References
-
- Hanahan D. Hallmarks of cancer: New dimensions. Cancer Discov. 2022;12:31–46. doi: 10.1158/2159-8290.CD-21-1059. - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

