EF1α, rather than CMV promoter, is suitable for luciferase tag expression in target cells for in vitro cytotoxicity assays of CAR-T cells
- PMID: 40777728
- PMCID: PMC12329255
- DOI: 10.1016/j.omtm.2025.101537
EF1α, rather than CMV promoter, is suitable for luciferase tag expression in target cells for in vitro cytotoxicity assays of CAR-T cells
Abstract
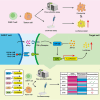
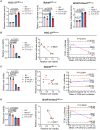
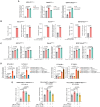
Precise assessment of the cytotoxic activity of engineered immune cell therapeutics, such as chimeric antigen receptor-engineered T (CAR-T) cells, is essential for their development and quality control. However, luciferase (Luc)-based viability assays, which evaluate target cell viability by overexpressing Luc tags and measuring chemiluminescent signals, may yield biased results depending on the promoter driving Luc expression. This study demonstrates that CAR-T cells can enhance cytomegalovirus (CMV) promoter-driven transcription in target cells via the interferon-gamma (IFN-γ)/nuclear factor κB (NF-κB) signaling pathway, leading to elevated Luc expression and a discrepancy between chemiluminescent signals and actual target cell death. These findings underscore the limitations of CMV promoters in functional protein overexpression systems in the context of engineered T cell killing of target cells due to their susceptibility to transcriptional interference. Statistical analyses indicate that Luc expression driven by the elongation factor-1 alpha (EF1α) promoter exhibits the highest concordance with flow cytometry-based quantification across three CAR-T cytotoxicity assay platforms, making it a more reliable choice for evaluating CAR-T cell cytotoxicity. This study highlights the necessity of selecting appropriate promoters to ensure accurate Luc-based cytotoxicity assessments and provides critical insights for standardizing detection methodologies in CAR-T cell evaluation.
Keywords: CAR-T cell therapy; CMV promoter; EF1α promoter; cytotoxicity assay; luciferase.
© 2025 The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
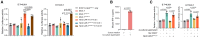
Similar articles
-
Chimeric antigen receptor (CAR) T-cell therapy for people with relapsed or refractory diffuse large B-cell lymphoma.Cochrane Database Syst Rev. 2021 Sep 13;9(9):CD013365. doi: 10.1002/14651858.CD013365.pub2. Cochrane Database Syst Rev. 2021. PMID: 34515338 Free PMC article.
-
B cell antigens: A key to optimizing CAR-T cell therapy.Int Rev Immunol. 2025 Jun 19:1-28. doi: 10.1080/08830185.2025.2515839. Online ahead of print. Int Rev Immunol. 2025. PMID: 40537997 Review.
-
In vitro machine learning-based CAR T immunological synapse quality measurements correlate with patient clinical outcomes.PLoS Comput Biol. 2022 Mar 18;18(3):e1009883. doi: 10.1371/journal.pcbi.1009883. eCollection 2022 Mar. PLoS Comput Biol. 2022. PMID: 35303007 Free PMC article. Clinical Trial.
-
Short-Term Memory Impairment.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31424720 Free Books & Documents.
-
[Volume and health outcomes: evidence from systematic reviews and from evaluation of Italian hospital data].Epidemiol Prev. 2013 Mar-Jun;37(2-3 Suppl 2):1-100. Epidemiol Prev. 2013. PMID: 23851286 Italian.
References
-
- Brentjens R.J., Latouche J.B., Santos E., Marti F., Gong M.C., Lyddane C., King P.D., Larson S., Weiss M., Rivière I., Sadelain M. Eradication of systemic B-cell tumors by genetically targeted human T lymphocytes co-stimulated by CD80 and interleukin-15. Nat. Med. 2003;9:279–286. doi: 10.1038/nm827. - DOI - PubMed
-
- Administration U.S.F.a.D., editor. Considerations for the Development of Chimeric Antigen Receptor (CAR) T Cell Products. Center for Biologics Evaluation and Research; 2024.
-
- Yamashita M., Kitano S., Aikawa H., Kuchiba A., Hayashi M., Yamamoto N., Tamura K., Hamada A. A novel method for evaluating antibody-dependent cell-mediated cytotoxicity by flowcytometry using cryopreserved human peripheral blood mononuclear cells. Sci. Rep. 2016;6 doi: 10.1038/srep19772. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

