Identification of a locus on Agropyron cristatum chromosome 6P that interacts with wheat genes to produce yellow-green leaves
- PMID: 40781275
- PMCID: PMC12333211
- DOI: 10.1186/s12870-025-07110-8
Identification of a locus on Agropyron cristatum chromosome 6P that interacts with wheat genes to produce yellow-green leaves
Abstract
Background: Wide hybridization is crucial for broadening the genetic basis of common wheat. Agropyron cristatum (2n = 4x = 28, PPPP), a wild relative of wheat, harbors numerous favorable genes for genetic improvement. The variability related to the expression of alien genes in different wheat backgrounds is a crucial factor that limits the effective utilization of these genes.
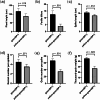
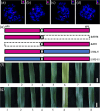
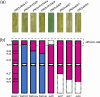
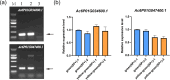
Results: In this study, the introduction of chromosome 6P from A. cristatum into different wheat backgrounds resulted in different leaf colors: green in plants with the Fukuho background and yellow‒green in plants with the Jimai 22 background. Genetic analysis suggested that yellow‒green leaves were caused by gene interactions between chromosome 6P and genes from the Jimai 22 background, which negatively affected agronomic traits. To determine the locus on chromosome 6P responsible for yellow‒green leaves, six wheat-A. cristatum deletion lines and five wheat-A. cristatum translocation lines were crossed with Jimai 22 to produce F1 progeny for leaf color investigation. We found that the F1 progeny carrying the short arm of chromosome 6P (6PS) presented yellow-green leaves, and the relevant locus was ultimately mapped to 6PS (0.81-1.00). A total of 50 A. cristatum genes related to chlorophyll catabolite reductase and chloroplast development were annotated within this interval.
Conclusions: A locus on chromosome 6P of A. cristatum that caused a yellow-green leaf in the Jimai 22 background was mapped to chromosome 6PS (0.81-1.00). This study provides valuable germplasm for the study of leaf color and guidance for the use of valuable genes on A. cristatum chromosome 6P.
Keywords: Agropyron cristatum; Chromosome 6P; Common wheat; Translocation line; Yellow–green leaf gene.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Our research did not involve any human or animal subjects, material, or data. Clinical trial number not applicable. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures


Similar articles
-
Deletion mapping and verification of an enhanced-grain number per spike locus from the 6PL chromosome arm of Agropyron cristatum in common wheat.Theor Appl Genet. 2019 Oct;132(10):2815-2827. doi: 10.1007/s00122-019-03390-5. Epub 2019 Jul 15. Theor Appl Genet. 2019. PMID: 31309244
-
Physical mapping of Agropyron cristatum chromosome 6P using deletion lines in common wheat background.Theor Appl Genet. 2016 May;129(5):1023-34. doi: 10.1007/s00122-016-2680-8. Epub 2016 Feb 26. Theor Appl Genet. 2016. PMID: 26920547
-
The effects of chromosome 6P on fertile tiller number of wheat as revealed in wheat-Agropyron cristatum chromosome 5A/6P translocation lines.Theor Appl Genet. 2015 May;128(5):797-811. doi: 10.1007/s00122-015-2466-4. Epub 2015 Feb 6. Theor Appl Genet. 2015. PMID: 25656149
-
The effect of sample site and collection procedure on identification of SARS-CoV-2 infection.Cochrane Database Syst Rev. 2024 Dec 16;12(12):CD014780. doi: 10.1002/14651858.CD014780. Cochrane Database Syst Rev. 2024. PMID: 39679851 Free PMC article.
-
What is the value of routinely testing full blood count, electrolytes and urea, and pulmonary function tests before elective surgery in patients with no apparent clinical indication and in subgroups of patients with common comorbidities: a systematic review of the clinical and cost-effective literature.Health Technol Assess. 2012 Dec;16(50):i-xvi, 1-159. doi: 10.3310/hta16500. Health Technol Assess. 2012. PMID: 23302507 Free PMC article.
References
-
- Shalygo N, Czarnecki O, Peter E, Grimm B. Expression of chlorophyll synthase is also involved in feedback-control of chlorophyll biosynthesis. Plant Mol Biol. 2009;71(4):425–36. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources

