HMGB1 as an emerging key modulator of bone remodeling: a narrative review
- PMID: 40781677
- PMCID: PMC12333100
- DOI: 10.1186/s13287-025-04543-8
HMGB1 as an emerging key modulator of bone remodeling: a narrative review
Abstract
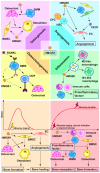
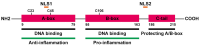
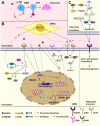
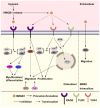
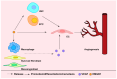
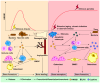
Bone is a dynamic tissue that undergoes microdamage and requires remodeling to replace old bone matrix. This process demands a tight coupling between bone resorption and bone formation. Aberrant internal environment can decouple these processes, reducing bone mass and compromising mechanical properties. High mobility group box-1 (HMGB1) is a multifunctional protein binding to advanced glycosylation end product-specific receptor (RAGE) and toll-like receptors (TLRs), regulating DNA repair, cytokine production, inflammation, cell proliferation, programmed cell death (PCD). Extracellular HMGB1 serves not only as a damage-associated molecular pattern (DAMP) that promotes cytokine storms and inflammatory cascade responses but also modulates osteogenesis, osteoclastogenesis and angiogenesis. Under physiological or acute trauma conditions, HMGB1 recruits osteoblasts, osteoclasts, and various immune cells to coordinate inflammation and immune responses, clearing pathogens and promoting bone repair. However, chronic HMGB1 overrelease strengthens osteoclastogenesis and bone resorption, leading to uncoupled bone remodeling and homeostatic imbalance. Thus, HMGB1 plays indispensable roles in bone metabolism and immune regulation. Current research on its involvement in bone remodeling remains incomplete and lacks systematic elucidation. This review aims to bridge this critical knowledge gap and provide a comprehensive reference for future investigations.
Keywords: Bone remodeling; HMGB1; Inflammation.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Not applicable. Consent for publication: Not applicable. Generative AI and AI-assisted technologies in the writing process: The authors declare that they have not use AI-generated work in this manuscript. Competing interests: The authors declare no competing interests.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

