The use of high-efficacy disease-modifying therapies in multiple sclerosis: recommendations from an expert Delphi consensus
- PMID: 40783885
- PMCID: PMC12336074
- DOI: 10.1007/s00415-025-13293-9
The use of high-efficacy disease-modifying therapies in multiple sclerosis: recommendations from an expert Delphi consensus
Abstract
Objective: To establish recommendations based on an expert consensus on the early and appropriate use of high-efficacy disease-modifying therapies (HE-DMTs) in the management of multiple sclerosis (MS) patients, based on current clinical evidence and real-world practice in Italy.
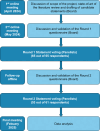
Material and methods: A Delphi panel comprising 65 neurologists from 54 Italian MS centers engaged in a two-round consensus process. Experts rated 43 statements across five domains: therapeutic goals, definitions of HE-DMT, MS patient profiling, and use of HE-DMT at diagnosis and later in MS course, using a 5-point Likert scale. A statement reached strong consensus if ≥80% of panelists agreed; whereas between 70% and 80% it was considered as moderate.
Results: In Round 2, 53 experts completed the survey on 43 statements. Strong consensus was achieved for 33 (76.7%), and moderate consensus for 6 (14.0%) statements. Experts strongly supported early HE-DMT initiation to prevent irreversible disability, endorsed a multidimensional definitions of treatment efficacy, and recommended personalized approaches based on clinical, radiological, and biomarker indicators. Consensus supported initiating HE-DMTs in patients with poor prognostic features and identified magnetic resonance imaging (MRI) activity, neurodegeneration markers, and suboptimal clinical response as specific factors requiring escalation to HE-DMTs.
Conclusion: This Italian Delphi underscores the importance of early, personalized HE-DMT use to optimize long-term outcomes in MS. The strong expert alignment reflects a paradigm shift toward proactive treatment and highlights actionable clinical, radiological, and biological indicators that should guide therapeutic decisions. These findings may support national policy changes and promote more equitable and evidence-based access to HE-DMTs across healthcare systems.
Keywords: Delphi; Multiple sclerosis; Treatment.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Conflicts of interest: The authors declare that they have no competing interests in relation to this work. Potential conflicts of interest outside the submitted work are as follows: M. Filippi is Editor-in-Chief of the Journal of Neurology, Associate Editor of Human Brain Mapping, Neurological Sciences, and Radiology, received compensation for consulting services from Alexion, Almirall, Biogen, Merck, Novartis, Roche, Sanofi, speaking activities from Bayer, Biogen, Celgene, Chiesi Italia SpA, Eli Lilly, Genzyme, Janssen, Merck-Serono, Neopharmed Gentili, Novartis, Novo Nordisk, Roche, Sanofi, Takeda, and TEVA, participation in Advisory Boards for Alexion, Biogen, Bristol-Myers Squibb, Merck, Novartis, Roche, Sanofi, Sanofi-Aventis, Sanofi-Genzyme, Takeda, scientific direction of educational events for Biogen, Merck, Roche, Celgene, Bristol-Myers Squibb, Lilly, Novartis, Sanofi-Genzyme, he receives research support from Biogen Idec, Merck-Serono, Novartis, Roche, the Italian Ministry of Health, the Italian Ministry of University and Research, and Fondazione Italiana Sclerosi Multipla. Maria Pia Amato has served on Scientific Advisory Boards for Biogen, Novartis, Roche, Merck, Sanofi Genzyme and Teva; has received speaker honoraria from Biogen, Merck, Sanofi Genzyme, Roche, Novartis and Teva; has received research grants for her Institution from Biogen, Merck, Sanofi Genzyme, Novartis and Roche. She is co-Editor of the Multiple Sclerosis Journal and Associate Editor of Frontiers in Neurology. Diego Centonze is an Advisory Board member or has given advice to Almirall, Bayer Schering, Biogen, GW Pharmaceuticals, Merck Serono, Novartis, Roche, Sanofi-Genzyme, and Teva; has received honoraria for speaking or consultation fees from Almirall, Bayer Schering, Biogen, GW Pharmaceuticals, Merck Serono, Novartis, Roche, Sanofi-Genzyme, and Teva; is the principal investigator in clinical trials for Bayer Schering, Biogen, Merck Serono, Mitsubishi, Novartis, Roche, Sanofi-Genzyme, and Teva. His preclinical and clinical research was supported by grants from Bayer Schering, Biogen Idec, Celgene, Merck Serono, Novartis, Roche, Sanofi-Genzyme and Teva. Paolo Gallo has been a consultant and member of Advisory Board for Biogen Italy, Sanofy, Merck, Almirall, Roche and Novartis, has received funding for travel and speaker honoraria from Merck-Serono, Biogen Idec, Sanofi, Novartis-Pharma, Roche, has received research support from Bayer, Biogen Italy, Merk, Sanofi, Roche, Novartis. Claudio Gasperini has served on Scientific Advisory Boards for Biogen, Novartis, Roche, Merck, Sanofi Genzyme; has received speaker honoraria from Biogen, Merck, Bayer, Sanofi Genzyme, Roche, Novartis, Almirall, Mylan. Matilde Inglese has received research grants from NIH, NMSS, FISM, EU. She is co-Editor of multiple sclerosis journal. She has received fees for participation in advisory boards from Biogen, Merck, Janssen, Novartis, Roche, Sanofi. Francesco Patti is involved in Advisor activity for: Alexion, Almirall, Bayer, Biogen, Bristol Meyers Squibb, Merck, Novartis, Sanofi and TEVA; in Speaking activity for: Almirall, Bayer, Biogen, Bristol Meyers Squibb, Merck, Sanofi and TEVA and in Research grants for: Biogen, Merck, Roche, ISS, FISM and MIUR FIR. Carlo Pozzilli is involved in scientific advisory boards for Biogen, Hoffmann-La Roche, Merck, Novartis, Janssen and Almirall; consulting and/or speaking fees from Almirall, Biogen, Bristol Myers, Janssen Hoffmann-La Roche, Merck, Novartis, and Biogen. Research support from Merck, Hoffman- La Roche, Novartis, Biogen. Paolo Preziosa received speaker honoraria from Roche, Biogen, Novartis, Merck, Bristol Myers Squibb, Genzyme, Horizon and Sanofi, he has received research support from Italian Ministry of Health and Fondazione Italiana Sclerosi Multipla. Maria Trojano has served on scientific advisory boards for Biogen, Novartis, Roche, the healthcare business of Merck KGaA, Darmstadt, Germany, Sandoz and Celgene (BMS); has received speaker honoraria from Biogen, Roche, Sanofi, the healthcare business of Merck KGaA, Darmstadt, Germany, Sandoz, Alexion and Novartis. Ethical approval: Not applicable.
Figures

References
-
- Filippi M, Bar-Or A, Piehl F, Preziosa P, Solari A, Vukusic S et al (2018) Multiple sclerosis. Nat Rev Dis Primers 4(1):43 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

