Development and validation of a machine learning model for predicting vulnerable carotid plaques using routine blood biomarkers and derived indicators: insights into sex-related risk patterns
- PMID: 40784899
- PMCID: PMC12337436
- DOI: 10.1186/s12933-025-02867-6
Development and validation of a machine learning model for predicting vulnerable carotid plaques using routine blood biomarkers and derived indicators: insights into sex-related risk patterns
Abstract
Background: Early detection of vulnerable carotid plaques is critical for stroke prevention. This study aimed to develop a machine learning model based on routine blood tests and derived indices to predict plaque vulnerability and assess sex-specific risk patterns across biomarker value ranges.
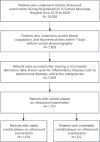
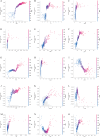
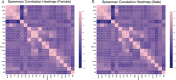
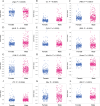
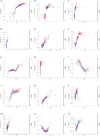
Methods: We retrospectively included 1701 hospitalized patients from Suzhou Municipal Hospital (2019-2020), selected from an initial cohort of 10,028 individuals. All patients underwent carotid ultrasound, with vulnerable plaques identified using predefined imaging criteria. A total of 30 laboratory variables-including blood count, coagulation, and biochemistry-were extracted, alongside derived indices such as triglyceride-glucose index (TyG), atherogenic index of plasma (AIP), neutrophil-to-lymphocyte ratio (NLR) and others. Features were standardized and selected based on statistical and clinical relevance. Five machine learning models were trained using a 7:3 train-test split and evaluated by cross-validation. Model performance was assessed using AUC, sensitivity, and specificity. The best model was interpreted using SHapley Additive exPlanations (SHAP) analysis. Sex differences were explored using Mann-Whitney U tests and restricted cubic spline (RCS) modeling across value intervals.
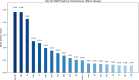
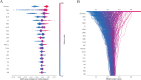
Results: The Random Forest model showed the highest predictive performance (AUC = 0.847; 95% CI 0.791-0.895; specificity = 89.4%; sensitivity = 64.2%). SHAP analysis identified gender, age, fibrinogen, NLR, creatinine, fasting blood glucose, uric acid to high-density lipoprotein ratio (UHR), TyG, systemic inflammation response index (SIRI), and lymphocyte count as top predictors. Significant sex-specific differences in SHAP values were observed for key biomarkers, including age, UHR, TyG, SIRI, and others. RCS modeling further revealed distinct sex-related patterns in plaque vulnerability across biomarker value ranges.
Conclusion: A Random Forest model integrating routine blood markers and derived indices accurately predicted vulnerable carotid plaques. The results underscore the importance of sex-specific risk assessment, highlighting differential effects of key biomarkers across genders and value intervals.
Keywords: Blood biomarkers; Machine learning; SHAP analysis; Sex differences; Vulnerable carotid plaque.
© 2025. The Author(s).
Conflict of interest statement
Supplementary information. Ethics approval and consent to participate: This study was a retrospective analysis approved by the Ethics Committee of Suzhou Municipal Hospital. Given the use of de-identified clinical data, the requirement for informed consent from individual participants was waived by the Ethics Committee. Competing interests: The authors declare no competing interests.
Figures

References
-
- Saba L, Cau R, Murgia A, Nicolaides AN, Wintermark M, Castillo M, Staub D, Kakkos SK, Yang Q, Paraskevas KI, et al. Carotid Plaque-RADS: a novel stroke risk classification system. JACC Cardiovasc Imaging. 2024;17(1):62–75. - PubMed
-
- Kopczak A, Schindler A, Bayer-Karpinska A, Koch ML, Sepp D, Zeller J, Strecker C, Hempel JM, Yuan C, Malik R, et al. Complicated carotid artery plaques as a cause of cryptogenic stroke. J Am Coll Cardiol. 2020;76(19):2212–22. - PubMed
-
- Saba L, Saam T, Jäger HR, Yuan C, Hatsukami TS, Saloner D, Wasserman BA, Bonati LH, Wintermark M. Imaging biomarkers of vulnerable carotid plaques for stroke risk prediction and their potential clinical implications. Lancet Neurol. 2019;18(6):559–72. - PubMed
-
- Johri AM, Herr JE, Li TY, Yau O, Nambi V. Novel ultrasound methods to investigate carotid artery plaque vulnerability. J Am Soc Echocardiogr Off Publ Am Soc Echocardiogr. 2017;30(2):139–48. - PubMed
-
- Schindler A, Schinner R, Altaf N, Hosseini AA, Simpson RJ, Esposito-Bauer L, Singh N, Kwee RM, Kurosaki Y, Yamagata S, et al. Prediction of stroke risk by detection of hemorrhage in carotid plaques: meta-analysis of individual patient data. JACC Cardiovasc Imaging. 2020;13(2 Pt 1):395–406. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

