Bimekizumab: The First FDA-Approved Dual IL-17A/IL-17F Inhibitor for Plaque Psoriasis - A Comprehensive Literature Review
- PMID: 40785801
- PMCID: PMC12335252
- DOI: 10.2147/PTT.S544166
Bimekizumab: The First FDA-Approved Dual IL-17A/IL-17F Inhibitor for Plaque Psoriasis - A Comprehensive Literature Review
Abstract
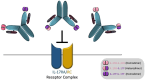
Psoriasis is a chronic inflammatory immune-mediated disease that affects 1-3% of the worldwide population. It is now known that interleukin IL-17F and IL-17A have a role in the pathophysiology of immune-mediated inflammatory disorders, such as psoriasis. According to recent data, neutralizing IL-17A and -17F together may be more effective than neutralizing IL-17A alone in treating psoriasis. Bimekizumab is a humanized IgG1 monoclonal antibody that selectively binds and diminishes the biological functions of both IL-17A and -17F. Current biologics for treating psoriasis lack complete skin clearance and reliable quick response. Bimekizumab's efficacy was evaluated in four randomized controlled trials involving around 2,200 patients with plaque psoriasis. The results showed that bimekizumab outperformed placebo, adalimumab, secukinumab, and ustekinumab, with higher response rates for both PASI 90 and PASI 100 at 16 weeks. Furthermore, Bimekizumab has shown superiority in direct comparative clinical studies (RCTs) performed. When compared to other biologics, Bimekizumab has shown a more rapid onset in terms of rapid response and better efficacy with a comparable safety profile as the most frequent adverse events include oral candidiasis, upper respiratory tract infections, and nasopharyngitis. These features of Bimekizumab provide advantages for the patients in term of better efficacy with rapid response and safe profile which ultimately affect the selection and treatment algorithm for management of plaque psoriasis.
Keywords: biologics; cytokine; dual inhibition; inflammatory disease; psoriasis.
© 2025 Al Alshaikh et al.
Conflict of interest statement
The authors declare no conflicts of interest in this work.
Figures
References
-
- Oliver R, Krueger JG, Glatt S, et al. Bimekizumab for the treatment of moderate‐to‐severe plaque psoriasis: efficacy, safety, pharmacokinetics, pharmacodynamics and transcriptomics from a phase IIa, randomized, double‐blind multicentre study. Br J Dermatol. 2022;186(4):652–663. doi: 10.1111/bjd.20827 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

