Ultrasound-Targeted Nanobubbles Codelivering NKP-1339 and miR-142-5p for Synergistic Mitochondrial Immunogenic Cell Death and PD-L1 Inhibition in Cancer Therapy
- PMID: 40785846
- PMCID: PMC12334082
- DOI: 10.34133/bmr.0232
Ultrasound-Targeted Nanobubbles Codelivering NKP-1339 and miR-142-5p for Synergistic Mitochondrial Immunogenic Cell Death and PD-L1 Inhibition in Cancer Therapy
Abstract
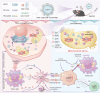
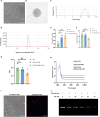
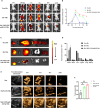
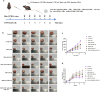
The combination of chemical immunotherapy and gene therapy holds great promise for malignant tumor treatment. Here, we developed an ultrasound-targeted liposome nanobubbles system (NKP-1339/miR-142-NBs) for precise codelivery of drugs and genes to treat esophageal squamous cell carcinoma (ESCC) with ultrasound-targeted microbubble destruction (UTMD). This study systematically investigated the system's therapeutic mechanisms-including mitochondrial dysfunction induction, immunogenic cell death (ICD), and antitumor immune activation-alongside its pharmacokinetics and targeting efficiency. In an ESCC mouse model, NKP-1339/miR-142-NBs combined with ultrasound markedly suppressed tumor growth (79.72% ± 0.1% vs. NB control 18.79% ± 1.29%) through NKP-1339 triggering ICD and miR-142-5p down-regulating programmed death-ligand 1 (PD-L1) expression, synergistically potentiating immune responses. Furthermore, we found that triggering ICD, including the exposure of calreticulin on the cell membrane, was related to altering mitochondrial fission dynamics in the ESCC cells. The down-regulation of PD-L1 expression by miR-142-5p reactivated CD8+ T cells by relieving programmed death-1 (PD-1)/PD-L1-mediated immunosuppression, enhancing immune memory and antitumor efficacy. Moreover, the UTMD technique enhanced the tumoral accumulation and penetration of nanobubbles, improving delivery specificity and minimizing off-target effects. This combined treatment strategy, including UTMD, provides a promising translational potential for ESCC therapy.
Copyright © 2025 Yafei Zhang et al.
Conflict of interest statement
Competing interests: The authors declare that they have no competing interests.
Figures
Similar articles
-
Ultrasound nanobubble-based combinational strategies of loaded miR-107-3p and CD133 Ab for anti-PD-L1 and anti-hepatocellular cancer stem cells.Int J Pharm. 2025 Feb 10;670:125140. doi: 10.1016/j.ijpharm.2024.125140. Epub 2025 Jan 3. Int J Pharm. 2025. PMID: 39756595
-
Interplay between tumor mutation burden and the tumor microenvironment predicts the prognosis of pan-cancer anti-PD-1/PD-L1 therapy.Front Immunol. 2025 Jul 24;16:1557461. doi: 10.3389/fimmu.2025.1557461. eCollection 2025. Front Immunol. 2025. PMID: 40777041 Free PMC article.
-
Evidence of immunogenic cell death (ICD) and ICD-dependent dendritic cell activation induced by extracorporeal photopheresis in patients with leukaemic forms of cutaneous T-cell lymphoma.Br J Dermatol. 2025 Jul 17;193(2):276-286. doi: 10.1093/bjd/ljaf102. Br J Dermatol. 2025. PMID: 40112181
-
Comparison of efficacy and safety of PD-1/PD-L1 combination therapy in first-line treatment of advanced NSCLC: an updated systematic review and network meta-analysis.Clin Transl Oncol. 2024 Oct;26(10):2488-2502. doi: 10.1007/s12094-024-03442-3. Epub 2024 Apr 16. Clin Transl Oncol. 2024. PMID: 38625495
-
Evaluation of efficacy, safety and underlying mechanism on Traditional Chinese medicine as synergistic agents for cancer immunotherapy: A preclinical systematic review and meta-analysis.J Ethnopharmacol. 2025 Feb 10;338(Pt 1):119035. doi: 10.1016/j.jep.2024.119035. Epub 2024 Nov 5. J Ethnopharmacol. 2025. PMID: 39510427
References
-
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–249. - PubMed
-
- Tacar O, Dass CR. Doxorubicin-induced death in tumour cells and cardiomyocytes: Is autophagy the key to improving future clinical outcomes? J Pharm Pharmacol. 2013;65(11):1577–1589. - PubMed
-
- Turajlic S, Swanton C. Metastasis as an evolutionary process. Science. 2016;352(6282):169–175. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

