An updated insight into the effect of β-adrenergic receptor antagonists (β-blockers) on respiratory function in asthma patients: a systematic review and meta-analysis
- PMID: 40786074
- PMCID: PMC12331582
- DOI: 10.3389/fphys.2025.1582740
An updated insight into the effect of β-adrenergic receptor antagonists (β-blockers) on respiratory function in asthma patients: a systematic review and meta-analysis
Abstract
Background: This study aimed to provide an updated assessment of the changes in respiratory function after β-adrenergic receptor antagonists (β-blockers) administration in asthma patients. The main assumption of the study was to use new methodological and statistical approaches not previously applied in this field in systematic reviews and meta-analyses.
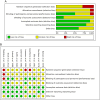
Methods: To select studies, PubMed/Medline, Embase, ClinicalTrials.gov, and Cochrane Library were searched. Additionally, Google Scholar was searched for gray literature. A systematic review and meta-analysis for forced expiratory volume in 1 second (FEV1) and peak expiratory flow rate in asthma patients after administration of cardio-selective and non-selective β-blockers compared to placebo or baseline was performed. We also assessed FEV1 after topical β-blocker application compared to baseline.
Results: An independent subgroup analysis demonstrated significantly higher FEV1 in the placebo group (standardized mean difference [SMD] =-0.74, 95% confidence interval [CI]: 1.15, -0.34, P = 0.0003) than in non-selective β-blockers. The test for subgroup differences indicates that there is a statistically significant subgroup effect among cardio-selective and non-selective β-blockers (P = 0.03, I 2 = 80%). We also showed a statistically significant decrease in FEV1 (SMD = -0.70, 95% CI: [-1.56 to -0.03], P = 0.04) after topical β-blocker application.
Conclusion: Patients with asthma may tolerate cardio-selective β-blockers better than non-selective β-blockers. The FEV1 value depends on the type of β-blocker used. Cardio-selective β-blockers may be cautiously considered in patients with asthma only when strong cardiovascular indications exist (such as heart failure with reduced ejection fraction or post-myocardial infarction) and with appropriate monitoring. At the same time, less risky therapeutic options should be chosen instead of topical β-blockers.
Systematic review registration: https://www.crd.york.ac.uk/prospero/, identifier 42024606876.
Keywords: FEV1; PEFR; asthma; beta-blockers; cardio-selective; non-selective; safety.
Copyright © 2025 Marko and Pawliczak.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


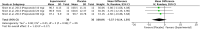
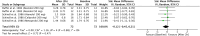
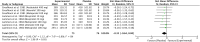

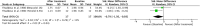
Similar articles
-
Beta-blockers in patients without heart failure after myocardial infarction.Cochrane Database Syst Rev. 2021 Nov 5;11(11):CD012565. doi: 10.1002/14651858.CD012565.pub2. Cochrane Database Syst Rev. 2021. PMID: 34739733 Free PMC article.
-
[Guidelines for the prevention and management of bronchial asthma (2024 edition)].Zhonghua Jie He He Hu Xi Za Zhi. 2025 Mar 12;48(3):208-248. doi: 10.3760/cma.j.cn112147-20241013-00601. Zhonghua Jie He He Hu Xi Za Zhi. 2025. PMID: 40050074 Chinese.
-
Beta-blockers for hypertension.Cochrane Database Syst Rev. 2007 Jan 24;(1):CD002003. doi: 10.1002/14651858.CD002003.pub2. Cochrane Database Syst Rev. 2007. Update in: Cochrane Database Syst Rev. 2012 Aug 15;(8):CD002003. doi: 10.1002/14651858.CD002003.pub3. PMID: 17253471 Updated.
-
Beta-blockers for hypertension.Cochrane Database Syst Rev. 2017 Jan 20;1(1):CD002003. doi: 10.1002/14651858.CD002003.pub5. Cochrane Database Syst Rev. 2017. PMID: 28107561 Free PMC article.
-
Interventions for infantile haemangiomas of the skin.Cochrane Database Syst Rev. 2018 Apr 18;4(4):CD006545. doi: 10.1002/14651858.CD006545.pub3. Cochrane Database Syst Rev. 2018. PMID: 29667726 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources

