Identification and quantification of GABAA R-α1-positive cells in the DCN of rats with behavioral evidence of noise-induced tinnitus
- PMID: 40791738
- PMCID: PMC12336005
- DOI: 10.1016/j.ibneur.2025.07.005
Identification and quantification of GABAA R-α1-positive cells in the DCN of rats with behavioral evidence of noise-induced tinnitus
Abstract
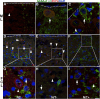
Tinnitus, the perception of a phantom sound, often occurs as a clinical sequela of auditory traumas. However, the underlying mechanisms of tinnitus are largely unknown. In our previous studies, we found more gamma-aminobutyric acid A receptor alpha 1 subunit (GABAAR-α1)-positive cells in the dorsal cochlear nucleus (DCN) after noise exposure, however, we were not able to identify the specific types of DCN cells that up-regulated GABAAR-α1 after the insult. In the current study, we used Nissl staining, Purkinje cell protein 4 (PCP4) and glutamic acid decarboxylase 67 (GAD67) immunolabeling to identify GABAAR-α1-positive cells in the DCN. Each type of GABAAR-α1-positive cells was quantified and statistically analyzed using immunostaining and Nissl staining according to their morphology, size and location in the DCN. GABAAR-α1-positive cartwheel cells, Golgi cells, as well as ML-stellate and vertical cells were confirmed by dual immunolabeling. In the DCN, the most common GABAAR-α1-positive cells were Golgi cells followed by vertical cells and cartwheel cells while very few other cells were GABAAR-α1-positive in all conditions. We found significantly more GABAAR-α1-positive Golgi cells in the DCN of noise-exposed rats without behavioral evidence of tinnitus compared to normal controls and noise-exposed rats with behavioral evidence of tinnitus. This heightened "context-dependent" inhibition may help to maintain a balanced neuronal network, preventing the potential for tinnitus-related hyperactivity in the auditory pathways.
Keywords: Acoustic trauma; Central auditory system; GABA receptor; Neurons; Tinnitus.
© 2025 The Authors.
Conflict of interest statement
Dr. Richard D. Kopke has financial interests in Otologic Pharmaceutics Inc. Drs. Xiaoping Du, Jianzhong Lu, Zachary Yokell, Matthew B. West, Mrs. Weihua Cheng and Mr. Don Nakmali have no conflict of interests.
Figures















References
LinkOut - more resources
Full Text Sources
Miscellaneous
