Insights into the role of MSLN-positive circulating tumor cell as an auxiliary diagnostic biomarker in epithelial ovarian cancer
- PMID: 40792273
- PMCID: PMC12336444
- DOI: 10.3389/fonc.2025.1563095
Insights into the role of MSLN-positive circulating tumor cell as an auxiliary diagnostic biomarker in epithelial ovarian cancer
Abstract
Background: Epithelial ovarian cancer (EOC) currently lacks highly specific biomarkers for clinical screening. This study aimed to identify and validate novel auxiliary diagnostic markers for EOC.
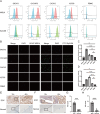
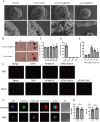
Methods: Through integrated analysis of transcriptome sequencing data and single-cell RNA sequencing from public databases, we identified mesothelin (MSLN) as an EOC-specific target. MSLN expression was subsequently validated in EOC cell lines and clinical specimens by flow cytometry, immunofluorescence, and immunohistochemistry. The capture efficacy of Pep@MNPs (Magnetic nanoparticles functionalised with EpCAM peptides) on EOC cells was verified by scanning electron microscopy, Prussian blue staining and cell spiked-blood capture experiments. In a prospective cohort of 35 patients with undiagnosed ovarian masses, we employed immunofluorescence staining to detect MSLN-positive circulating tumor cells (MSLN(+)CTCs) and assessed their diagnostic performance using receiver operating characteristic (ROC) analysis.
Results: MSLN was highly expressed in EOC cell line and tissues but lowly expressed in normal ovarian surface epithelial tissues. EOC cells can be captured by Pep@MNPs with high sensitivity and specificity. ROC curves analysis showed that MSLN(+)CTCs differentiated between benign and malignant lesions of the ovary with a sensitivity of 66.67% and a specificity of 95% (p = 0.0014), which was more specific than cancer antigen 125 (CA125) (sensitivity: 71.43%; specificity: 94.47%; p < 0.0001) and human epididymis protein 4 (HE4) (sensitivity: 84.62%; specificity: 89.47%; p = 0.0002). When MSLN(+)CTCs were combined with CA125, the sensitivity was 92.86% and the specificity was 94.74%, p < 0.0001, which greatly improved the diagnostic sensitivity while preserving high specificity.
Conclusions: MSLN(+)CTCs represent a highly specific auxiliary biomarker for differentiating benign and malignant ovarian lesions. The combination of MSLN(+)CTCs with CA125 provides an optimal balance between sensitivity and specificity, offering promising clinical utility for EOC diagnosis.
Keywords: EpCAM (CD326); MSLN; circulating tumor cells; diagnosis; epithelial ovarian cancer.
Copyright © 2025 Xu, Wang, Wu, Li, Wang, Zhang, Liu, Wang, Li, Hu, Liu and Yang.
Conflict of interest statement
Authors QL and MW were employed by the company Nanopep Biotech Corporation. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

