Aspirin, cardiovascular events, and major bleeding in older adults: extended follow-up of the ASPREE trial
- PMID: 40796244
- PMCID: PMC12596486
- DOI: 10.1093/eurheartj/ehaf514
Aspirin, cardiovascular events, and major bleeding in older adults: extended follow-up of the ASPREE trial
Abstract
Background and aims: Guidelines recommend against routine initiation of low-dose aspirin in older adults for primary prevention of atherosclerotic cardiovascular disease events. This study aimed to estimate long-term and post-trial effects of aspirin on major adverse cardiovascular events (MACE) and major haemorrhage using extended follow-up of participants from the ASPREE trial.
Methods: In-trial (2010-17) and post-trial (2017-22) data were analysed. At enrolment, participants were aged ≥70 years (≥65 years for US minorities) without prior cardiovascular events, dementia, or independence-limiting physical disability. Randomization was to daily low-dose aspirin or matching placebo for the 4.7 years of the trial.
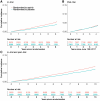
Results: Of the 19 114 participants randomized (9525 aspirin, 9589 placebo), 15 668 without in-trial MACE consented to post-trial follow-up. No long-term benefit of randomization to aspirin was observed for MACE for the entire in-trial and post-trial period [hazard ratio (HR) 1.04, 95% confidence interval (CI) .94, 1.15]. However, during the post-trial period (median 4.3 years), there was a higher rate of MACE (HR 1.17, 95% CI 1.01, 1.36) in those randomized to aspirin compared with placebo. Over the entire period, a higher rate of major haemorrhage was observed in the randomized aspirin group compared with placebo (HR 1.24, 95% CI 1.10, 1.39).
Conclusions: The present study provides novel evidence concerning long-term MACE and haemorrhage following aspirin use in initially healthy older adults. The finding of no long-term MACE benefit needs to be considered in clinical decision-making if aspirin is being considered for use in this context.
Keywords: Aspirin; Clinical trial; Coronary artery disease; Haemorrhage; Major adverse cardiovascular events; Observational follow-up; Stroke.
© The Author(s) 2025. Published by Oxford University Press on behalf of the European Society of Cardiology.
Figures




References
-
- Arnett DK, Blumenthal RS, Albert MA, Buroker AB, Goldberger ZD, Hahn EJ, et al. 2019 ACC/AHA guideline on the primary prevention of cardiovascular disease: a report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines. Circulation 2019;140:e596–646. 10.1161/CIR.0000000000000678 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

