Enhancing genome-scale metabolic models with kinetic data: resolving growth and citramalate production trade-offs in Escherichia coli
- PMID: 40799493
- PMCID: PMC12341681
- DOI: 10.1093/bioadv/vbaf166
Enhancing genome-scale metabolic models with kinetic data: resolving growth and citramalate production trade-offs in Escherichia coli
Abstract
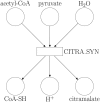
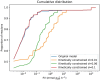
Summary: Metabolic models are valuable tools for analyzing and predicting cellular features such as growth, gene essentiality, and product formation. Among the various types of metabolic models, two prominent categories are constraint-based models and kinetic models. Constraint-based models typically represent a large subset of an organism's metabolic reactions and incorporate reaction stoichiometry, gene regulation, and constant flux bounds. However, their analyses are restricted to steady-state conditions, making it difficult to optimize competing objective functions. In contrast, kinetic models offer detailed kinetic information but are limited to a smaller subset of metabolic reactions, providing precise predictions for only a fraction of an organism's metabolism. To address these limitations, we proposed a hybrid approach that integrates these modeling frameworks by redefining the flux bounds in genome-scale constraint-based models using kinetic data. We applied this method to the constraint-based model of Escherichia coli, examining both its wild-type form and a genetically modified strain engineered for citramalate production. Our results demonstrate that the enriched model achieves more realistic reaction flux boundaries. Furthermore, by fixing the growth rate to a value derived from kinetic information, we resolved a flux bifurcation between growth and citramalate production in the modified strain, enabling accurate predictions of citramalate production rates.
Availability and implementation: The Python code generated for this work is available at: https://github.com/jlazaroibanezz/citrabounds.
© The Author(s) 2025. Published by Oxford University Press.
Conflict of interest statement
None declared.
Figures




Similar articles
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
Comparison of Two Modern Survival Prediction Tools, SORG-MLA and METSSS, in Patients With Symptomatic Long-bone Metastases Who Underwent Local Treatment With Surgery Followed by Radiotherapy and With Radiotherapy Alone.Clin Orthop Relat Res. 2024 Dec 1;482(12):2193-2208. doi: 10.1097/CORR.0000000000003185. Epub 2024 Jul 23. Clin Orthop Relat Res. 2024. PMID: 39051924
-
Short-Term Memory Impairment.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31424720 Free Books & Documents.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
Portion, package or tableware size for changing selection and consumption of food, alcohol and tobacco.Cochrane Database Syst Rev. 2015 Sep 14;2015(9):CD011045. doi: 10.1002/14651858.CD011045.pub2. Cochrane Database Syst Rev. 2015. PMID: 26368271 Free PMC article.
References
LinkOut - more resources
Full Text Sources

