Interhemispheric integration in the neural face perception network: Does stimulus location matter?
- PMID: 40800977
- PMCID: PMC12320007
- DOI: 10.1162/IMAG.a.17
Interhemispheric integration in the neural face perception network: Does stimulus location matter?
Abstract
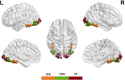
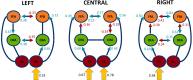
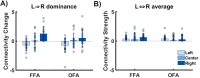
The neural mechanisms underlying hemispheric lateralization can be investigated using neuroimaging methods and modelling techniques. In some experiments, sensory information is initially presented exclusively to one hemisphere, for example, by displaying a visual stimulus in the periphery of the contralateral hemifield. This experimental design enables, among other things, a comparison of competing theories of interhemispheric integration (e.g., interhemispheric inhibition vs. interhemispheric recruitment). However, the underlying neural models for peripheral stimulation may differ from those for central stimulation and, therefore, may not adequately describe the mechanisms associated with typical, foveal stimulus processing. To address this question, the present functional magnetic resonance imaging (fMRI) study analysed the influence of stimulus location (peripheral vs. central) on neural network connectivity, particularly interhemispheric transfer, as determined by dynamic causal modelling (DCM), for a face perception task. Face and object images were presented either peripherally or centrally to a group of healthy volunteers (N= 17). By contrasting brain activations for faces against objects, we identified bilateral face-sensitive regions, such as the left and right fusiform face area (FFA) and the occipital face area (OFA). Additionally, we extracted the bilateral primary visual cortex (V1) as the input region for our neural models. We constructed five increasingly complex models that differed only in their modulatory connectivity. Bayesian model averaging (BMA) was employed to average the parameters across all models, enabling the calculation of interhemispheric transfer difference (i.e., left-to-right minus right-to-left modulatory connectivity parameter) and the strength of interhemispheric transfer between bilateral OFA and FFA regions. Our findings demonstrate that interhemispheric integration depends on stimulus location. Peripheral presentations of faces induce different connectivity patterns compared with centrally depicted faces. Specifically, we observed larger interhemispheric transfer differences for peripheral face stimuli compared with central stimuli. In conclusion, peripheral and central presentations of faces modulate the face processing network differently, with left and right visual field presentations yielding asymmetrical connectivity patterns. Since faces are preferentially processed via the fovea, the typical face processing network likely aligns more closely with activation patterns elicited by central stimuli. In contrast, connectivity patterns triggered by peripheral stimulation may represent an atypical processing style and cannot be directly compared with those activated by central stimuli.
Keywords: DCM; FFA; OFA; fMRI; face perception; interhemispheric transfer; lateralization; network; peripheral stimulation.
© 2025 The Authors. Published under a Creative Commons Attribution 4.0 International (CC BY 4.0) license.
Conflict of interest statement
The authors declare no competing interests.
Figures




References
LinkOut - more resources
Full Text Sources
