MicroRNA210 Suppresses Mitochondrial Metabolism and Promotes Microglial Activation in Neonatal Hypoxic-Ischemic Brain Injury
- PMID: 40801634
- PMCID: PMC12346595
- DOI: 10.3390/cells14151202
MicroRNA210 Suppresses Mitochondrial Metabolism and Promotes Microglial Activation in Neonatal Hypoxic-Ischemic Brain Injury
Abstract
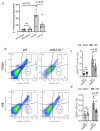
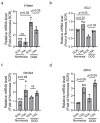
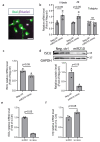
Neuroinflammation is the major contributor to the pathology of neonatal hypoxic-ischemic (HI) brain injury. Our previous studies have demonstrated that microRNA210 (miR210) inhibition with antisense locked nucleic acid (LNA) inhibitor mitigates neuroinflammation and provides neuroprotection after neonatal HI insult. However, the underlying mechanisms remain elusive. In the present study, using miR210 knockout (KO) mice and microglial cultures, we tested the hypothesis that miR210 promotes microglial activation and neuroinflammation through suppressing mitochondrial function in microglia after HI. Neonatal HI brain injury was conducted on postnatal day 9 (P9) wild-type (WT) and miR210 knockout (KO) mouse pups. We found that miR210 KO significantly reduced brain infarct size at 48 h and improved long-term locomotor functions assessed by an open field test three weeks after HI. Moreover, miR210 KO mice exhibited reduced IL1β levels, microglia activation and immune cell infiltration after HI. In addition, in vitro studies of microglia exposed to oxygen-glucose deprivation (OGD) revealed that miR210 inhibition with LNA reduced OGD-induced expression of Il1b and rescued OGD-mediated downregulation of mitochondrial iron-sulfur cluster assembly enzyme (ISCU) and mitochondrial oxidative phosphorylation activity. To validate the link between miR210 and microglia activation, isolated primary murine microglia were transfected with miR210 mimic or negative control. The results showed that miR210 mimic downregulated the expression of mitochondrial ISCU protein abundance and induced the expression of proinflammatory cytokines similar to the effect observed with ISCU silencing RNA. In summary, our results suggest that miR210 is a key regulator of microglial proinflammatory activation through reprogramming mitochondrial function in neonatal HI brain injury.
Keywords: hypoxia–ischemia; microglia; mitochondrial dysfunction; neuroinflammation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
The Neuroprotective Mechanisms of PPAR-γ: Inhibition of Microglia-Mediated Neuroinflammation and Oxidative Stress in a Neonatal Mouse Model of Hypoxic-Ischemic White Matter Injury.CNS Neurosci Ther. 2024 Nov;30(11):e70081. doi: 10.1111/cns.70081. CNS Neurosci Ther. 2024. PMID: 39496476 Free PMC article.
-
PHLDA1 contributes to hypoxic ischemic brain injury in neonatal rats via inhibiting FUNDC1-mediated mitophagy.Acta Pharmacol Sin. 2024 Sep;45(9):1809-1820. doi: 10.1038/s41401-024-01292-x. Epub 2024 May 15. Acta Pharmacol Sin. 2024. PMID: 38750074
-
MicroRNA-210 Downregulates ISCU and Induces Mitochondrial Dysfunction and Neuronal Death in Neonatal Hypoxic-Ischemic Brain Injury.Mol Neurobiol. 2019 Aug;56(8):5608-5625. doi: 10.1007/s12035-019-1491-8. Epub 2019 Jan 17. Mol Neurobiol. 2019. PMID: 30656514 Free PMC article.
-
Remote Ischemic Postconditioning Improve Cerebral Ischemia-Reperfusion Injury Induced Cognitive Dysfunction through Suppressing Mitochondrial Apoptosis in Hippocampus via TK/BK/B2R-Mediated PI3K/AKT.Mol Neurobiol. 2025 Aug;62(8):10652-10669. doi: 10.1007/s12035-025-04864-y. Epub 2025 Apr 14. Mol Neurobiol. 2025. PMID: 40229456 Free PMC article.
-
Potential therapeutic targets for hypoxic-ischemic brain injury based on mitochondrial reprogramming mechanism.Neuroscience. 2025 Sep 13;583:76-83. doi: 10.1016/j.neuroscience.2025.07.043. Epub 2025 Aug 2. Neuroscience. 2025. PMID: 40752813 Review.
References
-
- Lee A.C., Kozuki N., Blencowe H., Vos T., Bahalim A., Darmstadt G.L., Niermeyer S., Ellis M., Robertson N.J., Cousens S., et al. Intrapartum-related neonatal encephalopathy incidence and impairment at regional and global levels for 2010 with trends from 1990. Pediatr. Res. 2013;74((Suppl. S1)):50–72. doi: 10.1038/pr.2013.206. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

