Biallelic variants in DNAJC7 cause familial amyotrophic lateral sclerosis with the TDP-43 pathology
- PMID: 40802071
- PMCID: PMC12350594
- DOI: 10.1007/s00401-025-02899-y
Biallelic variants in DNAJC7 cause familial amyotrophic lateral sclerosis with the TDP-43 pathology
Abstract
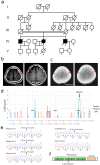
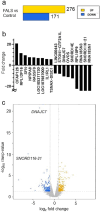
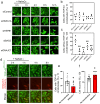
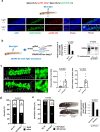
Amyotrophic lateral sclerosis (ALS) is a fatal neurodegenerative disorder characterized by the progressive degeneration of motor neurons. ALS pathology primarily involves the failure of protein quality control mechanisms, leading to the accumulation of misfolded proteins, particularly TAR DNA-binding protein 43 (TDP-43). TDP-43 aggregation is a central pathological feature of ALS. Maintaining protein homeostasis is critical and facilitated by heat shock proteins (HSPs), particularly the HSP40 family, which includes co-chaperones such as DNAJC7. Here, we report a family with three siblings affected by ALS who carry a homozygous c.518dupC frameshift variant in DNAJC7, a member of the HSP40 family. All three patients exhibited progressive muscle weakness, limb atrophy, bulbar palsy, and respiratory failure. Pathological examination revealed degeneration of both upper and lower motor neurons, with phosphorylated TDP-43-positive neuronal cytoplasmic inclusions in the frontal and temporal cortices. Immunoblot analysis were consistent with a type B pattern of phosphorylated TDP-43 in the precentral gyrus. Immunohistochemistry and RNA sequencing analyses demonstrated a substantial reduction in DNAJC7 expression at both the protein and RNA levels in affected brain regions. In a TDP-43 cell model, DNAJC7 knockdown impaired the disassembly of TDP-43 following arsenite-induced stress, whereas DNAJC7 overexpression suppressed the assembly and promoted the disassembly of arsenite-induced TDP-43 condensates. Furthermore, in a zebrafish ALS model, dnajc7 knockdown resulted in increased TDP-43 aggregation in motor neurons and reduced survival. To the best of our knowledge, this study provides the first evidence linking biallelic loss-of-function variants in DNAJC7 to familial ALS with TDP-43 pathology.
Keywords: DNAJC7; Amyotrophic lateral sclerosis; Heat shock protein; Live-cell imaging; TDP-43; Zebrafish disease model.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Conflict of interest: The authors declare no potential conflict of interest. Ethical approval: The study was conducted in accordance with the Ethical Guidelines for Human Genome/Gene Analysis Research and the Guidelines for Medical and Health Research Involving Human Subjects, as set forth by the Japanese government. The use of human biological samples was approved by the Ethics Review committees of Okayama University, the National Hospital Organisation Minami-Okayama Medical Centre, Zikei Hospital, and the Tokyo Metropolitan Institute of Medical Science. Written informed consent was obtained from the family members of the deceased.
Figures


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

