Elucidating the functional role of the novel BdP50 protein and extracellular vesicles in the human erythrocyte infection by Babesia divergens
- PMID: 40802835
- PMCID: PMC12370190
- DOI: 10.1371/journal.pntd.0013401
Elucidating the functional role of the novel BdP50 protein and extracellular vesicles in the human erythrocyte infection by Babesia divergens
Abstract
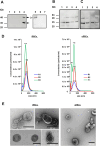
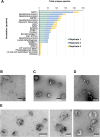
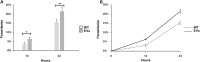
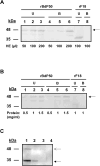
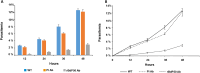
Babesia divergens is a blood-borne parasite that invades, replicates within and destroys red blood cells (RBCs) during its asexual life cycle, causing babesiosis in humans and cattle. This study focuses on BdP50, a putative B. divergens glycosylphosphatidylinositol-anchored protein involved in the parasite life cycle. BdP50 is found on the surface of B. divergens invasive parasites (merozoites) as well as on extracellular vesicles (Bd-derived EVs). These EVs are secreted by parasites cultured in fresh human RBCs and, in addition to BdP50, are enriched in human and parasite proteins, including proteins related to the parasite invasion process. BdP50 binds to RBCs and could mediate interactions of free merozoites and Bd-derived EVs with the host cell. Anti-BdP50 antibodies support this by blocking the BdP50 protein and inhibiting up to 88% of merozoite entry into naïve RBCs. This reinforces the role of BdP50 in parasite-host cell interactions and invasion. However, the inhibitory effect of BdP50 antibodies begins to gradually decrease slightly several hours after invasion, leading to a progressive increase in B. divergens infected RBCs over time. Consistent with these findings, our in vitro de novo infection assays showed that Bd-derived EVs, in addition to promoting parasite propagation, display proteins such as BdP50 that mimic the merozoite surface to likely attenuate the blocking effect of antibodies, thereby ensuring the parasite survival during subsequent rounds of invasion and growth. Given the role of BdP50 and Bd-derived EVs in the B. divergens life cycle, this study could have future implications for developing new approaches to interfere with parasite invasion proteins and Bd-derived EVs functions.
Copyright: © 2025 Gonzalez et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

