Clinical characteristics and long-term outcomes of 101 patients with urea cycle disorders in China
- PMID: 40804416
- PMCID: PMC12351844
- DOI: 10.1186/s13023-025-03985-w
Clinical characteristics and long-term outcomes of 101 patients with urea cycle disorders in China
Abstract
Background: Urea cycle disorders (UCDs) are a group of rare genetic metabolic disorders characterized by hyperammonemia, which can lead to neurological damage, systemic complications, and even death. Understanding UCDs' clinical features and progression in the Chinese population will fill research gaps and benefit patients globally.
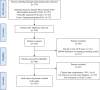
Methods: This retrospective study evaluated the clinical, biochemical, genetic characteristics, and long-term outcomes in 101 Chinese patients with six subtypes of UCDs between 2007 and 2024. Data were collected from medical records and analyzed.
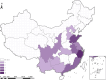
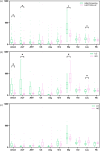
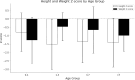
Results: The overall survival rate was 93.0% among UCD patients. An equal gender ratio was observed in ornithine transcarbamylase deficiency. Newborn screening (NBS) was conducted in this cohort, and 57.0% of patients were diagnosed through NBS. Neurological and gastrointestinal symptoms were the most common. Symptoms often appeared within the first year, especially in the first month. Arginine was the most frequently used treatment, with glycerol phenylbutyrate often used as a nitrogen scavenger in severe cases. Biochemical analysis showed subtype-specific differences, including notable declines in leucine and glycine on low-protein diets. Genetic analysis revealed a wide distribution of mutations, with few hotspots and 17 newly identified mutations. Clinically diagnosed patients had worse outcomes than those diagnosed via newborn screening.
Conclusion: This study is the first to describe the clinical features and long-term outcomes of UCDs in a large sample of Chinese patients, highlighting the importance of newborn screening for early diagnosis and improved treatment outcomes.
Keywords: Genetic mutations; Hyperammonemia; Long-term outcomes; Newborn screening; Urea cycle disorders.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: All the participants signed written informed consent and consented to participate in this investigation. The research protocol and process were approved by the Ethical Committee of The Children’s Hospital of Zhejiang University School of Medicine (reference number: 2021-IRB-292). All methods used in the present study were performed following the relevant guidelines and regulations. Consent for publication: Informed consent was obtained from all patients or their legal guardians. Competing interests: The authors declare that they have no competing interests.
Figures

Similar articles
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
Severity-adjusted evaluation of initial dialysis on short-term health outcomes in urea cycle disorders.Mol Genet Metab. 2024 Sep-Oct;143(1-2):108566. doi: 10.1016/j.ymgme.2024.108566. Epub 2024 Aug 19. Mol Genet Metab. 2024. PMID: 39299137
-
Ornithine Transcarbamylase Deficiency.2013 Aug 29 [updated 2022 May 26]. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2025. 2013 Aug 29 [updated 2022 May 26]. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2025. PMID: 24006547 Free Books & Documents. Review.
-
Systemic treatments for metastatic cutaneous melanoma.Cochrane Database Syst Rev. 2018 Feb 6;2(2):CD011123. doi: 10.1002/14651858.CD011123.pub2. Cochrane Database Syst Rev. 2018. PMID: 29405038 Free PMC article.
-
The current social status in adult patients with urea cycle disorders in Japan.Mol Genet Metab. 2025 Aug;145(4):109185. doi: 10.1016/j.ymgme.2025.109185. Epub 2025 Jul 1. Mol Genet Metab. 2025. PMID: 40618446
References
-
- Deng K, Zhu J, Yu E, Xiang L, Yuan X, Yao Y, Li X, Liu H. Incidence of inborn errors of metabolism detected by tandem mass spectrometry in China: a census of over seven million newborns between 2016 and 2017. J Med Screen. 2021;28(3):223–9. 10.1177/0969141320973690. - PubMed
-
- Husson MC, Schiff M, Fouilhoux A, Cano A, Dobbelaere D, Brassier A, Mention K, Arnoux JB, Feillet F, Chabrol B, et al. Efficacy and safety of i.v. sodium benzoate in urea cycle disorders: a multicentre retrospective study. Orphanet J Rare Dis. 2016;11(1): 127. 10.1186/s13023-016-0513-0. - PMC - PubMed
-
- Posset R, Garbade SF, Boy N, Burlina AB, Dionisi-Vici C, Dobbelaere D, Garcia-Cazorla A, de Lonlay P, Teles EL, Vara R, et al. Transatlantic combined and comparative data analysis of 1095 patients with urea cycle disorders-A successful strategy for clinical research of rare diseases. J Inherit Metab Dis. 2019;42(1):93–106. 10.1002/jimd.12031. - PMC - PubMed
-
- Kido J, Matsumoto S, Mitsubuchi H, Endo F, Nakamura K. Early liver transplantation in neonatal-onset and moderate urea cycle disorders may lead to normal neurodevelopment. Metab Brain Dis. 2018;33(5):1517–23. 10.1007/s11011-018-0259-6. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

