YAP/TAZ Promote GLUT1 Expression and Are Associated with Prognosis in Endometrial Cancer
- PMID: 40805250
- PMCID: PMC12345884
- DOI: 10.3390/cancers17152554
YAP/TAZ Promote GLUT1 Expression and Are Associated with Prognosis in Endometrial Cancer
Abstract
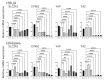
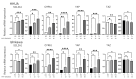
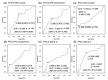
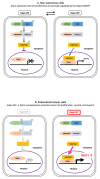
Background/Objectives: Yes-associated protein (YAP) and transcriptional coactivator with PDZ-binding motif (TAZ) function as effectors in the Hippo pathway and have attracted attention due to their association with tumor formation. Glucose transporter (GLUT) proteins also contribute to the proliferation of cancer cells. In this study, we investigated the effect of YAP/TAZ on GLUT1 expression in endometrial carcinoma, as well as the clinical relevance and prognostic value of YAP/TAZ. Methods: The effects of YAP and TAZ knockdown and YAP overexpression on GLUT1 expression in human endometrial carcinoma-derived HHUA and Ishikawa cells were evaluated using RT-qPCR. In addition, we performed immunohistochemical expression of 100 tissue samples of diagnosed endometrial carcinoma. Based on staining intensity and the percentage of positively stained tumor cells, the immunoreactivity score was calculated, which ranged from 0 to 12. Results: YAP/TAZ were identified as important factors in the regulation of GLUT1 expression in HHUA and Ishikawa cells. In addition, a significant correlation (progression-free survival p < 0.05) was observed between TAZ and GLUT1 expression in tissues from endometrial carcinoma patients, and nuclear expression of TAZ was associated with poor prognosis (p < 0.05). Conclusions: YAP/TAZ promote tumor growth via GLUT1. Therapeutic targeting of YAP/TAZ could therefore be useful in the development of future treatments.
Keywords: GLUT1; TAZ; YAP; endometrial cancer; hippo pathway.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures





Similar articles
-
The polarity protein Par3 enhances renal cell carcinoma metastasis via YAP/TAZ activation.Cancer Biol Med. 2025 Jul 8;22(7):812-31. doi: 10.20892/j.issn.2095-3941.2024.0297. Cancer Biol Med. 2025. PMID: 40626835 Free PMC article.
-
YAP/TAZ-TEAD activity promotes the malignant transformation of cervical intraepithelial neoplasia through enhancing the characteristics and Warburg effect of cancer stem cells.Apoptosis. 2024 Aug;29(7-8):1198-1210. doi: 10.1007/s10495-023-01935-0. Epub 2024 Mar 29. Apoptosis. 2024. PMID: 38553612 Free PMC article.
-
Injinoryeong-San attenuates metabolic dysfunction-associated steatohepatitis via regulation of YAP/TAZ-signaling pathway.J Ethnopharmacol. 2025 Jul 17;353(Pt A):120292. doi: 10.1016/j.jep.2025.120292. Online ahead of print. J Ethnopharmacol. 2025. PMID: 40683423
-
Complex roles of Hippo-YAP/TAZ signaling in hepatocellular carcinoma.J Cancer Res Clin Oncol. 2023 Nov;149(16):15311-15322. doi: 10.1007/s00432-023-05272-2. Epub 2023 Aug 22. J Cancer Res Clin Oncol. 2023. PMID: 37608027 Free PMC article. Review.
-
Exploring Hippo YAP/TAZ Signaling: A Novel Avenue for Cardiovascular Disorders.Cell Biol Int. 2025 Sep;49(9):1079-1101. doi: 10.1002/cbin.70052. Epub 2025 Jul 14. Cell Biol Int. 2025. PMID: 40654305 Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

