In-Depth Characterization of L1CAM+ Extracellular Vesicles as Potential Biomarkers for Anti-CD20 Therapy Response in Relapsing-Remitting Multiple Sclerosis
- PMID: 40806340
- PMCID: PMC12347089
- DOI: 10.3390/ijms26157213
In-Depth Characterization of L1CAM+ Extracellular Vesicles as Potential Biomarkers for Anti-CD20 Therapy Response in Relapsing-Remitting Multiple Sclerosis
Abstract
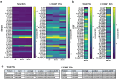
The effective suppression of inflammation using disease-modifying therapies is essential in the treatment of multiple sclerosis (MS). Anti-CD20 monoclonal antibodies are commonly used long-term as maintenance therapies, largely due to the lack of reliable biomarkers to guide dosing and evaluate treatment response. However, prolonged use increases the risk of infections and other immune-mediated side effects. The unique ability of brain-derived blood extracellular vesicles (EVs) to cross the blood-brain barrier and reflect the central nervous system (CNS) immune status has sparked interest in their potential as biomarkers. This study aimed to assess whether blood-derived L1CAM+ EVs could serve as biomarkers of treatment response to rituximab (RTX) in patients with relapsing-remitting MS (RRMS). Serum samples (n = 25) from the baseline (month 0) and after 6 months were analyzed from the RTX arm of the ongoing randomized clinical trial OVERLORD-MS (comparing anti-CD20 therapies in RRMS patients) and were compared with serum samples from healthy controls (n = 15). Baseline cerebrospinal fluid (CSF) samples from the same study cohort were also included. EVs from both serum and CSF samples were characterized, considering morphology, size, and concentration, using transmission electron microscopy (TEM) and nanoparticle tracking analysis (NTA). The immunophenotyping of EV surface receptors was performed using flow cytometry with the MACSPlex exosome kit, while label-free quantitative proteomics of EV protein cargo was conducted using a proximity extension assay (PEA). TEM confirmed the presence of EVs with the expected round morphology with a diameter of 50-150 nm. NTA showed significantly higher concentrations of L1CAM+ EVs (p < 0.0001) in serum total EVs and EBNA1+ EVs (p < 0.01) in serum L1CAM+ EVs at baseline (untreated) compared to in healthy controls. After six months of RTX therapy, there was a significant reduction in L1CAM+ EV concentration (p < 0.0001) and the downregulation of TNFRSF13B (p = 0.0004; FC = -0.49) in serum total EVs. Additionally, non-significant changes were observed in CD79B and CCL2 levels in serum L1CAM+ EVs at baseline compared to in controls and after six months of RTX therapy. In conclusion, L1CAM+ EVs in serum showed distinct immunological profiles before and after rituximab treatment, underscoring their potential as dynamic biomarkers for individualized anti-CD20 therapy in MS.
Keywords: anti-CD20 therapy; brain-derived blood exosomes; characterization of L1CAM+ EVs; multiple sclerosis (MS); rituximab; treatment-response biomarkers.
Conflict of interest statement
K.-M.M. has served on a scientific advisory board for Alexion, received speaker honoraria from Biogen, Lundbeck, Novartis, and Roche, and has participated in clinical trials organized by Biogen, Merck, Novartis, Roche, and Sanofi. O.T. has participated in advisory boards and received speaker honoraria from Biogen, Merck, Novartis, Teva, Roche, Sanofi, and Bristol Myers Squibb and has participated in clinical trials organized by Merck, Novartis, Roche, and Sanofi. The other authors declare no conflicts of interest.
Figures

Similar articles
-
Extracellular Vesicle-Derived MicroRNAs as a Biomarker for Therapeutic Response in Multiple Sclerosis.Neurol Neuroimmunol Neuroinflamm. 2025 Jul;12(4):e200420. doi: 10.1212/NXI.0000000000200420. Epub 2025 Jun 19. Neurol Neuroimmunol Neuroinflamm. 2025. PMID: 40537080 Free PMC article.
-
Rituximab for relapsing-remitting multiple sclerosis.Cochrane Database Syst Rev. 2013 Dec 6;2013(12):CD009130. doi: 10.1002/14651858.CD009130.pub3. Cochrane Database Syst Rev. 2013. PMID: 24310855 Free PMC article.
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
Central nervous system-derived extracellular vesicles: the next generation of neural circulating biomarkers?Transl Neurodegener. 2024 Jun 19;13(1):32. doi: 10.1186/s40035-024-00418-9. Transl Neurodegener. 2024. PMID: 38898538 Free PMC article. Review.
-
Most L1CAM Is not Associated with Extracellular Vesicles in Human Biofluids and iPSC-Derived Neurons.Mol Neurobiol. 2025 Aug;62(8):10427-10442. doi: 10.1007/s12035-025-04909-2. Epub 2025 Apr 10. Mol Neurobiol. 2025. PMID: 40210837 Free PMC article.
References
-
- Walton C., King R., Rechtman L., Kaye W., Leray E., Marrie R.A., Robertson N., La Rocca N., Uitdehaag B., van der Mei I., et al. Rising prevalence of multiple sclerosis worldwide: Insights from the Atlas of MS, third edition. Mult. Scler. 2020;26:1816–1821. doi: 10.1177/1352458520970841. - DOI - PMC - PubMed
-
- Fernández-Fournier M., López-Molina M., Torres Iglesias G., Botella L., Chamorro B., Laso-García F., Puertas I., Tallón Barranco A., Otero-Ortega L., Frank-García A., et al. Antibody Content against Epstein–Barr Virus in Blood Extracellular Vesicles Correlates with Disease Activity and Brain Volume in Patients with Relapsing–Remitting Multiple Sclerosis. Int. J. Mol. Sci. 2023;24:14192. doi: 10.3390/ijms241814192. - DOI - PMC - PubMed
-
- Bjornevik K., Cortese M., Healy B.C., Kuhle J., Mina M.J., Leng Y., Elledge S.J., Niebuhr D.W., Scher A.I., Munger K.L., et al. Longitudinal analysis reveals high prevalence of Epstein-Barr virus associated with multiple sclerosis. Science. 2022;375:296–301. doi: 10.1126/science.abj8222. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

