Investigating Neuroprotective Effects of Berberine on Mitochondrial Dysfunction and Autophagy Impairment in Parkinson's Disease
- PMID: 40806472
- PMCID: PMC12346907
- DOI: 10.3390/ijms26157342
Investigating Neuroprotective Effects of Berberine on Mitochondrial Dysfunction and Autophagy Impairment in Parkinson's Disease
Abstract
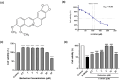
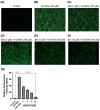
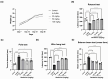
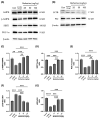
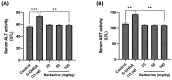
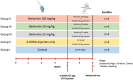
Parkinson's disease (PD) is a common neurodegenerative disorder with substantial global impact. Although current therapies can provide symptomatic relief, they are often associated with high costs and adverse effects. Natural compounds with a history of traditional medicinal use have emerged as promising alternatives. In this study, we investigated the therapeutic potential and underlying mechanisms of berberine in both cellular and animal models of PD. In vitro, SH-SY5Y cells exposed to 6-hydroxydopamine (6-OHDA) exhibited decreased viability and increased oxidative stress, both of which were significantly alleviated by berberine treatment based on cell viability assays and DCFH-DA staining. Western blot analysis revealed that berberine modulated the AMPK-PGC-1α-SIRT1 signaling pathway and restored the expression of autophagy-related proteins LC3B and P62, suggesting that berberine could improve mitochondrial function and autophagy balance. In vivo studies using a 6-OHDA-induced PD mouse model further confirmed these effects, showing that berberine could improve motor function and lead to molecular changes consistent with in vitro studies. Additionally, safety evaluations indicated no significant hepatotoxicity based on AST and ALT levels. Body weight also remained stable throughout treatment. Collectively, our findings suggest that berberine can not only alleviate PD-related symptoms but also target key pathological mechanisms, supporting its potential as a therapeutic candidate for PD and other neurodegenerative diseases.
Keywords: Parkinson’s disease; autophagy impairment; berberine; mitochondria dysfunction; natural product.
Conflict of interest statement
Author Jin-Hyeob Ryu was employed by the company BIOCHESTRA Co. Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest
Figures

Similar articles
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
Exercise Ameliorates Dysregulated Mitochondrial Fission, Mitochondrial Respiration, and Neuronal Apoptosis in Parkinson's Disease Mice via the Irisin/AMPK/SIRT1 Pathway.Mol Neurobiol. 2025 Jul;62(7):8843-8856. doi: 10.1007/s12035-025-04801-z. Epub 2025 Mar 6. Mol Neurobiol. 2025. PMID: 40048058
-
Berberine's paradox in Neurodegeneration: Therapeutic promise and safety challenges in Parkinson's disease.Neuropharmacology. 2025 Nov 1;278:110555. doi: 10.1016/j.neuropharm.2025.110555. Epub 2025 Jun 6. Neuropharmacology. 2025. PMID: 40482960 Review.
-
Neuroprotective effects of quercetin on motor impairments and anxiety-like behaviors in a rat model of Parkinson's disease.Behav Brain Res. 2025 Sep 13;493:115692. doi: 10.1016/j.bbr.2025.115692. Epub 2025 Jun 10. Behav Brain Res. 2025. PMID: 40505972
-
Cholinesterase inhibitors for dementia with Lewy bodies, Parkinson's disease dementia and cognitive impairment in Parkinson's disease.Cochrane Database Syst Rev. 2012 Mar 14;2012(3):CD006504. doi: 10.1002/14651858.CD006504.pub2. Cochrane Database Syst Rev. 2012. PMID: 22419314 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

