Artificial Gametogenesis and In Vitro Spermatogenesis: Emerging Strategies for the Treatment of Male Infertility
- PMID: 40806512
- PMCID: PMC12347267
- DOI: 10.3390/ijms26157383
Artificial Gametogenesis and In Vitro Spermatogenesis: Emerging Strategies for the Treatment of Male Infertility
Abstract
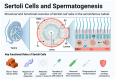
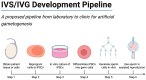
Male-factor infertility accounts for approxiamately half of all infertility cases globally, yet therapeutic options remain limited for individuals with no retrievable spermatozoa, such as those with non-obstructive azoospermia (NOA). In recent years, artificial gametogenesis has emerged as a promising avenue for fertility restoration, driven by advances in two complementary strategies: organotypic in vitro spermatogenesis (IVS), which aims to complete spermatogenesis ex vivo using native testicular tissue, and in vitro gametogenesis (IVG), which seeks to generate male gametes de novo from pluripotent or reprogrammed somatic stem cells. To evaluate the current landscape and future potential of these approaches, a narrative, semi-systematic literature search was conducted in PubMed and Scopus for the period January 2010 to February 2025. Additionally, landmark studies published prior to 2010 that contributed foundational knowledge in spermatogenesis and testicular tissue modeling were reviewed to provide historical context. This narrative review synthesizes multidisciplinary evidence from cell biology, tissue engineering, and translational medicine to benchmark IVS and IVG technologies against species-specific developmental milestones, ranging from rodent models to non-human primates and emerging human systems. Key challenges-such as the reconstitution of the blood-testis barrier, stage-specific endocrine signaling, and epigenetic reprogramming-are discussed alongside critical performance metrics of various platforms, including air-liquid interface slice cultures, three-dimensional organoids, microfluidic "testis-on-chip" devices, and stem cell-derived gametogenic protocols. Particular attention is given to clinical applicability in contexts such as NOA, oncofertility preservation in prepubertal patients, genetic syndromes, and reprocutive scenarios involving same-sex or unpartnered individuals. Safety, regulatory, and ethical considerations are critically appraised, and a translational framework is outlined that emphasizes biomimetic scaffold design, multi-omics-guided media optimization, and rigorous genomic and epigenomic quality control. While the generation of functionally mature sperm in vitro remains unachieved, converging progress in animal models and early human systems suggests that clinically revelant IVS and IVG applications are approaching feasibility, offering a paradigm shift in reproductive medicine.
Keywords: ethical considerations; in vitro gametogenesis; in vitro spermatogenesis; male infertility; organoids; pluripotent stem cells.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
A comparison of spermatogenesis between flies and men-conserved processes of male gamete production.Hum Reprod Update. 2025 Aug 13:dmaf018. doi: 10.1093/humupd/dmaf018. Online ahead of print. Hum Reprod Update. 2025. PMID: 40802929
-
Building a human pluripotent stem cell-based gonadal niche: improving in vitro systems with in vivo insights.Hum Reprod Update. 2025 Jun 28:dmaf012. doi: 10.1093/humupd/dmaf012. Online ahead of print. Hum Reprod Update. 2025. PMID: 40580487
-
Artificial gametes: a systematic review of biological progress towards clinical application.Hum Reprod Update. 2015 May-Jun;21(3):285-96. doi: 10.1093/humupd/dmv001. Epub 2015 Jan 21. Hum Reprod Update. 2015. PMID: 25609401
-
Improving sperm selection strategies for assisted reproduction through closing the knowledge gap in sperm maturation mechanics.Hum Reprod Open. 2025 Jul 3;2025(3):hoaf040. doi: 10.1093/hropen/hoaf040. eCollection 2025. Hum Reprod Open. 2025. PMID: 40698010 Free PMC article. Review.
References
-
- Kaltsas A., Dimitriadis F., Zachariou D., Zikopoulos A., Symeonidis E.N., Markou E., Tien D.M.B., Takenaka A., Sofikitis N., Zachariou A. From Diagnosis to Treatment: Comprehensive Care by Reproductive Urologists in Assisted Reproductive Technology. Medicina. 2023;59:1835. doi: 10.3390/medicina59101835. - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

