Microtubule-Targeting Agents: Advances in Tubulin Binding and Small Molecule Therapy for Gliomas and Neurodegenerative Diseases
- PMID: 40806780
- PMCID: PMC12347508
- DOI: 10.3390/ijms26157652
Microtubule-Targeting Agents: Advances in Tubulin Binding and Small Molecule Therapy for Gliomas and Neurodegenerative Diseases
Abstract
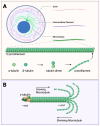
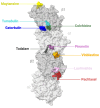
Microtubules play a key role in cell division and cell migration. Thus, microtubule-targeting agents (MTAs) are pivotal in cancer therapy due to their ability to disrupt cell division microtubule dynamics. Traditionally divided into stabilizers and destabilizers, MTAs are increasingly being repurposed for central nervous system (CNS) applications, including brain malignancies such as gliomas and neurodegenerative diseases like Alzheimer's and Parkinson's. Microtubule-stabilizing agents, such as taxanes and epothilones, promote microtubule assembly and have shown efficacy in both tumour suppression and neuronal repair, though their CNS use is hindered by blood-brain barrier (BBB) permeability and neurotoxicity. Destabilizing agents, including colchicine-site and vinca domain binders, offer potent anticancer effects but pose greater risks for neuronal toxicity. This review highlights the mapping of nine distinct tubulin binding pockets-including classical (taxane, vinca, colchicine) and emerging (tumabulin, pironetin) sites-that offer new pharmacological entry points. We summarize the recent advances in structural biology and drug design, enabling MTAs to move beyond anti-mitotic roles, unlocking applications in both cancer and neurodegeneration for next-generation MTAs with enhanced specificity and BBB penetration. We further discuss the therapeutic potential of combination strategies, including MTAs with radiation, histone deacetylase (HDAC) inhibitors, or antibody-drug conjugates, that show synergistic effects in glioblastoma models. Furthermore, innovative delivery systems like nanoparticles and liposomes are enhancing CNS drug delivery. Overall, MTAs continue to evolve as multifunctional tools with expanding applications across oncology and neurology, with future therapies focusing on optimizing efficacy, reducing toxicity, and overcoming therapeutic resistance in brain-related diseases.
Keywords: cancer; cytoskeleton; neurodegeneration; tubulin.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
RGN6024 Is a Brain-Penetrant, Small-Molecule Tubulin Destabilizer for the Treatment of Glioblastoma.Mol Cancer Ther. 2025 Aug 1;24(8):1129-1144. doi: 10.1158/1535-7163.MCT-24-1208. Mol Cancer Ther. 2025. PMID: 40397474
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
Systemic treatments for metastatic cutaneous melanoma.Cochrane Database Syst Rev. 2018 Feb 6;2(2):CD011123. doi: 10.1002/14651858.CD011123.pub2. Cochrane Database Syst Rev. 2018. PMID: 29405038 Free PMC article.
-
Design and synthesis of novel 4-aryl-2-benzoyl-imidazoles as colchicine binding site inhibitors.Eur J Med Chem. 2025 Nov 15;298:118021. doi: 10.1016/j.ejmech.2025.118021. Epub 2025 Aug 4. Eur J Med Chem. 2025. PMID: 40763661
-
Recent Advances in Microtubule Targeting Agents for Cancer Therapy.Molecules. 2025 Aug 8;30(16):3314. doi: 10.3390/molecules30163314. Molecules. 2025. PMID: 40871468 Review.
References
-
- Cash A.D., Aliev G., Siedlak S.L., Nunomura A., Fujioka H., Zhu X., Raina A.K., Vinters H.V., Tabaton M., Johnson A.B., et al. Microtubule reduction in Alzheimer’s disease and aging is independent of tau filament formation. Am. J. Pathol. 2003;162:1623–1627. doi: 10.1016/S0002-9440(10)64296-4. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

