Real-World Effectiveness of Rosuvastatin-Ezetimibe Single Pill (Rovazet®) in Korean Dyslipidemia Patients
- PMID: 40807110
- PMCID: PMC12347665
- DOI: 10.3390/jcm14155480
Real-World Effectiveness of Rosuvastatin-Ezetimibe Single Pill (Rovazet®) in Korean Dyslipidemia Patients
Abstract
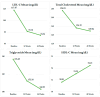
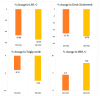
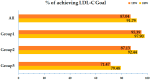
Background: Fixed-dose combinations of rosuvastatin and ezetimibe are increasingly used in clinical practice, but real-world data on their effectiveness and safety in large populations remain limited. Methods: This prospective, single-group, open-label, non-interventional observational study was conducted in the Republic of Korea to evaluate the effectiveness and safety of Rovazet® (a fixed-dose combination of rosuvastatin and ezetimibe). Patients were prospectively enrolled from 235 institutions (50 general hospitals and 185 private clinics) as part of routine clinical practice over a five-year period. Lipid profiles and medication compliance questionnaire results were collected at baseline, 12 weeks, and 24 weeks of treatment. Results: A total of 5527 patients with dyslipidemia, the majority were men (53.0%), and the mean age was 60.4 years. Rovazet® significantly reduced low-density lipoprotein cholesterol (LDL-C) by 23.5% at 12 weeks (from 117.47 ± 50.65 mg/dL to 81.14 ± 38.20 mg/dL; p < 0.0001) and by 27.4% at 24 weeks (from 117.47 ± 50.65 mg/dL to 74.52 ± 33.36 mg/dL; p < 0.0001). Total cholesterol was significantly reduced by 17.7% at 12 weeks and by 19.8% at 24 weeks. Rovazet® treatment reduced triglycerides by 4.1% at 12 weeks and by 7.2% at 24 weeks. High-density lipoprotein cholesterol increased by 4.5% at 12 weeks and by 7.9% at 24 weeks following Rovazet® treatment. These changes in lipid profiles were consistent, regardless of cardiovascular risk profiles. By 24 weeks of treatment with Rovazet®, 91.8% of patients had reached their target LDL-C goals. Adverse drug reactions were reported in 2.81% of patients, most of which were minor, indicating that Rovazet® was well tolerated. Conclusions: Rovazet® was effective in improving lipid profiles and well tolerated in Korean adults with dyslipidemia.
Keywords: dyslipidemia; ezetimibe; observational study; rosuvastatin; safety.
Conflict of interest statement
The authors declare no conflicts of interest. This study was supported by a research grant from HK inno.N Corporation, which was involved in the study design statistical planning, and data collection and analysis. The study conduct, interpretation of results, and manuscript preparation were carried out independently by the authors without sponsor influence.
Figures
Similar articles
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
Adding Ezetimibe to High-Intensity Statin at the Time of Admission in Patients With Acute Coronary Syndrome: Results From the Optimization of Low-Density Lipoprotein Cholesterol After Acute Coronary Syndrome (OLA) Study.Cureus. 2025 Aug 6;17(8):e89513. doi: 10.7759/cureus.89513. eCollection 2025 Aug. Cureus. 2025. PMID: 40777069 Free PMC article.
-
Efficacy and safety of combination therapy Ezetimibe 10/rosuvastatin 40 in Egyptian patients at very high risk of atherosclerotic cardiovascular disease.Egypt Heart J. 2025 Jun 11;77(1):59. doi: 10.1186/s43044-025-00655-x. Egypt Heart J. 2025. PMID: 40498290 Free PMC article.
-
A Systematic Review of PCSK9 Inhibitors Alirocumab and Evolocumab.J Manag Care Spec Pharm. 2016 Jun;22(6):641-653q. doi: 10.18553/jmcp.2016.22.6.641. J Manag Care Spec Pharm. 2016. PMID: 27231792 Free PMC article.
-
The quantity, quality and findings of network meta-analyses evaluating the effectiveness of GLP-1 RAs for weight loss: a scoping review.Health Technol Assess. 2025 Jun 25:1-73. doi: 10.3310/SKHT8119. Online ahead of print. Health Technol Assess. 2025. PMID: 40580049 Free PMC article.
References
-
- Tsao C.W., Aday A.W., Almarzooq Z.I., Anderson C.A.M., Arora P., Avery C.L., Baker-Smith C.M., Beaton A.Z., Boehme A.K., Buxton A.E., et al. Heart Disease and Stroke Statistics-2023 Update: A Report from the American Heart Association. Circulation. 2023;147:e93–e621. doi: 10.1161/CIR.0000000000001123. - DOI - PMC - PubMed
-
- Mach F., Baigent C., Catapano A.L., Koskinas K.C., Casula M., Badimon L., Chapman M.J., De Backer G.G., Delgado V., Ference B.A., et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk. Eur. Heart J. 2020;41:111–188. doi: 10.1093/eurheartj/ehz455. - DOI - PubMed
-
- Grundy S.M., Stone N.J., Bailey A.L., Beam C., Birtcher K.K., Blumenthal R.S., Braun L.T., de Ferranti S., Faiella-Tommasino J., Forman D.E., et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139:e1082–e1143. doi: 10.1161/cir.0000000000000625. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

