Protein dysregulation during Leishmania infantum infection in anti-TNF immunosuppressed mice revealed through quantitative proteomics analysis of extracellular vesicles
- PMID: 40808943
- PMCID: PMC12343234
- DOI: 10.3389/fimmu.2025.1634080
Protein dysregulation during Leishmania infantum infection in anti-TNF immunosuppressed mice revealed through quantitative proteomics analysis of extracellular vesicles
Abstract
Introduction: Visceral leishmaniasis (VL) occurs more frequently in immunosuppressed individuals, especially those undergoing immunosuppressive drug therapy for an autoimmune disease. In those receiving TNF antagonist therapy (anti-TNF), the course of VL is more severe and the response to traditional leishmanicidal treatments, such as antimonials (Sb), is often reduced. This effect of anti-TNF treatment is observed in our immunosuppressed-mouse model of VL. In this model, we compared anti-TNF immunosuppression with no immunosuppression before and after VL treatment with Sb.
Methods: Serum-derived extracellular vesicles (EVs) were analyzed through label-free quantitative proteomics to identify proteins involved in both VL severity and the impact of anti-TNF immunosuppression on treatment outcome.
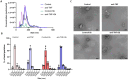
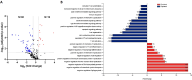
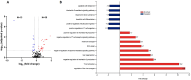
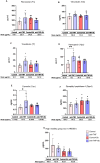
Results: In total, 223 dysregulated proteins were found in the pre-treatment groups, the majority of which, such as vitronectin, haemopexin or caveolin-1, were downregulated in the anti-TNF samples. In contrast, 173 proteins were identified in the Sb-treatment groups, most of which were found enriched in the anti-TNF plus treatment samples (anti-TNF+Sb) including fibronectin, transferrin, vitronectin and dipeptidyl peptidase-4. These differentially-expressed proteins were associated with pathways related to the immune system, liver regeneration, and ion transport.
Conclusion: Our findings have useful implications for the clinical management of VL patients under anti-TNF immunosuppression.
Keywords: LFQ proteomics analyses; TNF antagonist; antimonials; biomarkers; extracellular vesicles; immunosuppression; quantitative proteomics; visceral leishmaniasis.
Copyright © 2025 Bernardo, Montero-Calle, Solana, Lozano-Rendal, Torres, Sánchez, Barderas, Moreno and Carrillo.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Gradoni L, Lopez-Velez R, Mokni M. Manual on case management and surveillance of the leishmaniases in the WHO European Region. Geneva, Switzerland: World Health Organisation; (2017).
-
- Kurizky PS, Marianelli FF, Cesetti MV, Damiani G, Sampaio RNR, Goncalves LMT, et al. A comprehensive systematic review of leishmaniasis in patients undergoing drug-induced immunosuppression for the treatment of dermatological, rheumatological and gastroenterological diseases. Rev Inst Med Trop Sao Paulo. (2020) 62:e28. doi: 10.1590/s1678-9946202062028, PMID: - DOI - PMC - PubMed
-
- Bosch-Nicolau P, Ubals M, Salvador F, Sanchez-Montalva A, Aparicio G, Erra A, et al. Leishmaniasis and tumor necrosis factor alpha antagonists in the Mediterranean basin. A switch in clinical expression. PloS Negl Trop Dis. (2019) 13:e0007708. doi: 10.1371/journal.pntd.0007708, PMID: - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

