Genetic alterations affect immune contexture of non-small cell lung cancer: Ukrainian study
- PMID: 40809430
- PMCID: PMC12345293
- DOI: 10.3389/fmed.2025.1558016
Genetic alterations affect immune contexture of non-small cell lung cancer: Ukrainian study
Abstract
Introduction: Although the role of various genetic alterations was highlighted among factors affecting the response to immunotherapy in non-small cell lung cancer (NSCLC), the relations between oncogenic driver variants and changes in the cancer immunity cycle are still unclear.
Aim: The study aimed to discover the links between the molecular and immune context of NSCLC.
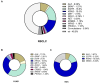
Materials and methods: This cohort study included 254 cases of NSCLC (193 Lung Adenocarcinomas) (LUAD; 76%), and 61 squamous cell carcinomas (SCC; 24%), with pathology reports and next-generation sequencing (NGS) data available. First, the rate and spectrum of genetic alterations were assessed in the Ukrainian cohort. Second, we uncovered the relationship between the oncogenic driver mutations and PD-L1 expression in NSCLC. Finally, T-cytotoxic lymphocytes (CD8+) and tumor-associated macrophages (CD163+) were evaluated in samples with EGFR and KRAS mutations, ALK rearrangements and LUAD with no genetic findings. Immune desert, immune excluded and inflamed types of tumor immune microenvironment (TME) were defined according to the cancer immunity cycle.
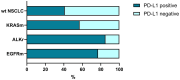
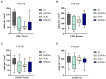
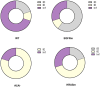
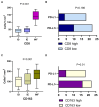
Results: More than half (52%) of the observed NSCLC cases harbored single (48.03%) or concomitant (3.94%) genetic alterations in oncogenes. The Ukrainian cohort demonstrated a high rate of EGFR (18.5%) and ALK rearrangements (9.4%) with a relatively moderate frequency of KRAS mutations (16.9%). NSCLC tumors with alterations in EGFR and ALK demonstrated a high incidence of PD-L1 expression and specific immune contexture. The number of CD8+ cells varied significantly between oncogene-driven and wild-type LUAD (p = 0.019). Non-oncogene-addicted NSCLC demonstrated the prevalence of Inflamed TME rich in CD163+ macrophages. In contrast, over half of EGFR mutant LUAD cases possessed immune desert TME type, while ALK-rearranged and KRAS mutant NSCLC showed mostly immune excluded TME.
Conclusion: The high rate of PD-L1 expression in NSCLC driven by EGFR and ALK alterations was accompanied by a prevalence of low immunogenicity with a shift toward ID TME in EGFR mutant tumors and IE TME in ALK-rearranged and KRAS mutant NSCLC. Further discovery of mechanisms affecting tumor immune contexture is needed for tailoring patient management in line with particular mechanisms of immune evasion.
Keywords: adenocarcinoma; cancer immunity cycle; genetic alterations; immune contexture; non-small cell lung cancer; oncogenes.
Copyright © 2025 Kozakov, Kobyliak, Livshun, Seleznov, Koshyk, Matvieieva, Shparyk, Kolesnik, Moskalenko, Vynnychenko, Moskalenko, Kropyvko, Khmel, Shkarupii and Sulaieva.
Conflict of interest statement
BS was employed by company Audubon Bioscience. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures
Similar articles
-
Frequency of driver oncogenic alterations in NSCLC and estimated indoor radon exposure in Europe (RADON EUROPE study).Ther Adv Med Oncol. 2025 Jun 26;17:17588359251351449. doi: 10.1177/17588359251351449. eCollection 2025. Ther Adv Med Oncol. 2025. PMID: 40585875 Free PMC article.
-
Targeted therapy for advanced anaplastic lymphoma kinase (<I>ALK</I>)-rearranged non-small cell lung cancer.Cochrane Database Syst Rev. 2022 Jan 7;1(1):CD013453. doi: 10.1002/14651858.CD013453.pub2. Cochrane Database Syst Rev. 2022. PMID: 34994987 Free PMC article.
-
Unveiling unique protein and phosphorylation signatures in lung adenocarcinomas with and without ALK, EGFR, and KRAS genetic alterations.Mol Oncol. 2025 Jul 7. doi: 10.1002/1878-0261.70091. Online ahead of print. Mol Oncol. 2025. PMID: 40621945
-
Association of PD-L1 expression with driver gene mutations and clinicopathological characteristics in non-small cell lung cancer: A real-world study of 10 441 patients.Thorac Cancer. 2024 Apr;15(11):895-905. doi: 10.1111/1759-7714.15244. Epub 2024 Mar 8. Thorac Cancer. 2024. PMID: 38456253 Free PMC article.
-
PD-L1 expression in advanced NSCLC: Insights into risk stratification and treatment selection from a systematic literature review.Lung Cancer. 2017 Oct;112:200-215. doi: 10.1016/j.lungcan.2017.08.005. Epub 2017 Aug 10. Lung Cancer. 2017. PMID: 29191596
References
-
- Fitzmaurice C, Abate D, Abbasi N, Abbastabar H, Abd-Allah F, Abdel-Rahman O, et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2017: a systematic analysis for the global burden of disease study. JAMA Oncol. (2019) 5:1749–68. doi: 10.1001/JAMAONCOL.2019.2996, PMID: - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

