What Is Regulating Chironomid Populations? The Influence of Food Supply and Interference Competition on Development and Mortality in Chironomus riparius
- PMID: 40809829
- PMCID: PMC12344276
- DOI: 10.1002/ece3.71949
What Is Regulating Chironomid Populations? The Influence of Food Supply and Interference Competition on Development and Mortality in Chironomus riparius
Abstract
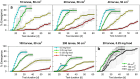
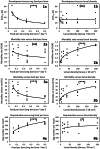
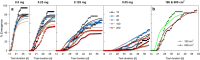
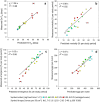
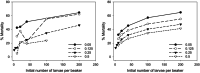
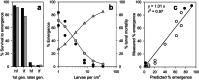
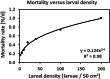
Density-dependent processes are important for a fundamental understanding of population regulation, as well as for understanding responses to and recovery from stressors. While exploitative competition is well-studied, interference competition is rather difficult to investigate, but it has been regularly observed to occur in many aquatic insect populations. We conducted laboratory experiments with the non-biting midge Chironomus riparius (Diptera: Chironomidae) to investigate the impact of different combinations of food supply and larval densities on development and mortality at a constant temperature of 20°C. The chosen two-factorial experimental design allowed a separate evaluation of exploitative (food) and interference (mortality) competition across a gradient of larval densities. The use of different vessel sizes between 50 cm2 and 600 cm2 made it possible to quantify the functional response at different food densities. To test mechanistic explanations for the statistically significant empirical relationships found in this study and to predict density-dependent processes, we used a dynamic process-oriented modeling approach. We extended a recently developed DEB-IBM full life cycle model for C. riparius and successfully applied it under variable food conditions at the population level under laboratory conditions. Our study showed that chironomid development and reproduction are primarily dependent on food supply, whereas larval density drives the density-dependent mortality rate. The interaction of food availability and interference competition determined the effective mortality over time. Killing by conspecifics was the most likely mechanism responsible for the intraspecific mortality of the larval stages. Combining data generated using a tailor-made experimental design with a mechanistic model provided insights into and quantified regulation mechanisms of chironomid populations, allowing future uses of this information in the context of population-level risk assessment from exposure to chemicals.
Keywords: Diptera; cannibalism; density dependence; dynamic energy budget model DEB; individual‐based model IBM; interference competition.
© 2025 The Author(s). Ecology and Evolution published by British Ecological Society and John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflicts of interest. However, Syngenta uses population models to assess the impact of chemicals on target and non‐target species in the context of pesticide regulation.
Figures

References
-
- Accolla, C. , Schmolke A., Vaugeois M., and Galic N.. 2023. “Density‐Dependent Population Regulation in Freshwater Fishes and Small Mammals: A Literature Review and Insights for Ecological Risk Assessment.” Integrated Environmental Assessment and Management 20, no. 5: 1225–1236. - PubMed
-
- Åkerblom, N. , and Goedkoop W.. 2003. “Stable Isotopes and Fatty Acids Reveal That Chironomus riparius Feeds Selectively on Added Food in Standardized Toxicity Tests.” Environmental Toxicology and Chemistry 22, no. 7: 1473–1480. - PubMed
-
- Armitage, P. D. 1995. “Chironomidae as Food.” In The Chironomidae: Biology and Ecology of Non‐Biting Midges, edited by Armitage P. D., Cranston P. S., and Pinder L. C. V., 423–435. Springer.
-
- Azevedo‐Pereira, H. M. , Abreu S. N., Lemos M. F., and Soares A. M.. 2012. “Bioaccumulation and Elimination of Waterborne Mercury in the Midge Larvae, Chironomus riparius Meigen (Diptera: Chironomidae).” Bulletin of Environmental Contamination and Toxicology 89: 245–250. - PubMed
-
- Beaty, T. V. 1995. The Use of Chironomus riparius (Diptera: Chironomidae) in Benthic Toxicity Tests and Its Response to Selenium. Doctoral Dissertation. Virginia Tech.
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources

