In vivo dynamic imaging reveals the oviduct as a leaky peristaltic pump in transporting a preimplantation embryo toward pregnancy
- PMID: 40809967
- PMCID: PMC12339303
- DOI: 10.1364/BOE.565065
In vivo dynamic imaging reveals the oviduct as a leaky peristaltic pump in transporting a preimplantation embryo toward pregnancy
Abstract
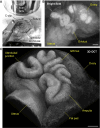
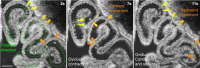
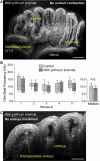
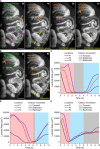
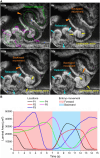
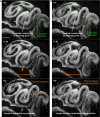
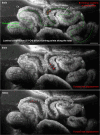
The mammalian oviduct (also called the fallopian tube) is an essential organ for natural pregnancy. As one of its major functions, the oviduct transports preimplantation embryos to the uterus for implantation. This is a critical process, and abnormalities are responsible for a range of reproductive disorders, such as tubal ectopic pregnancy and infertility, whose etiologies are unclear. For transporting embryos, the oviduct is fundamentally a tubular mechanical pump with motile cilia lining the luminal epithelium and smooth muscle surrounding the mucosa wall. Although bidirectional movement of embryos has been observed during the transport process, how the oviduct produces this type of embryo movement remains unknown. Understanding this pumping mechanism is vital to identifying the functional causes of oviduct-related reproductive disorders, but answering this question requires dynamic imaging of the transport process in its native environment, which is difficult to achieve in mammalian models. Here, we use optical coherence tomography and apply in vivo dynamic 3D imaging of the mouse oviduct to uncover the oviduct pumping mechanism in transporting preimplantation embryos toward pregnancy. By inhibiting the oviduct smooth muscle contraction, we first show that the oviduct muscular activity drives the bidirectional embryo movement. We then present a quantitative assessment of the oviduct contraction wave. This analysis, together with the embryo movement information, indicates that the forward movement of embryos is produced by peristalsis, while the backward embryo movement is generated by a suction process driven by the oviduct relaxation at earlier contraction sites, showing a leaky peristaltic pump. Finally, we reveal how the net displacement of embryos is created under this pumping mechanism, which effectively transports embryos toward the uterus. This work elucidates, for the first time, the oviduct pumping mechanism in transporting preimplantation embryos, paving the way for understanding the biomechanics of the mammalian oviduct.
© 2025 Optica Publishing Group.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Avilés M., Coy P., Rizos D., “The oviduct: A key organ for the success of early reproductive events,” Animal Frontiers 5(1), 25–31 (2015). 10.2527/af.2015-0005 - DOI
Associated data
LinkOut - more resources
Full Text Sources
Research Materials
