Basigin links altered skeletal stem cell lineage dynamics with glucocorticoid-induced bone loss and impaired angiogenesis
- PMID: 40817087
- PMCID: PMC12356905
- DOI: 10.1038/s41467-025-62881-w
Basigin links altered skeletal stem cell lineage dynamics with glucocorticoid-induced bone loss and impaired angiogenesis
Abstract
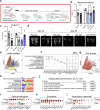
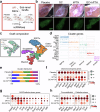
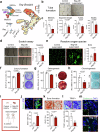
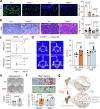
Glucocorticoid (GC) induced osteoporosis (GIOP) and osteonecrosis remain a significant health issue with few approved therapies. Here, we investigate the cellular and molecular processes by which GCs affect osteogenesis and angiogenesis. We find that GC treatment reduces bone mass through decreased bone formation by skeletal stem cells (SSCs). Concomitantly, endothelial cells increase in number but display distorted phenotypical features. Transplantation studies of SSCs combined with molecular analysis by single cell RNA-sequencing and functional testing of primary human cells tie GC-induced skeletal changes to altered stem cell differentiation dynamics. This in turn perpetuates reduced osteogenesis and vascular malformation through direct SSC-endothelial crosstalk mediated at least in part by Basigin. The genetic deletion of Basigin in the skeletal lineage as well as antibody-mediated blockade of Basigin during GC treatment prevents bone loss. Intriguingly, when administered to 2-year-old mice, anti-Basigin therapy reinstates bone remodeling to significantly improve bone mass. These findings provide therapeutic vantage points for GIOP and potentially other conditions associated with bone loss.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures



References
-
- Chang, C., Greenspan, A. & Gershwin, M. E. The pathogenesis, diagnosis and clinical manifestations of steroid-induced osteonecrosis. J. Autoimmun.110, 102460 (2020). - PubMed
-
- van Staa, T. P., Leufkens, H. G. M. & Cooper, C. The epidemiology of corticosteroid-induced osteoporosis: a meta-analysis. Osteoporos. Int.13, 777–787 (2002). - PubMed
MeSH terms
Substances
Grants and funding
- S10 OD028493/OD/NIH HHS/United States
- EDUC4-12792/California Institute for Regenerative Medicine (CIRM)
- na/Prostate Cancer Foundation (PCF)
- R00 AG049958/AG/NIA NIH HHS/United States
- R00 AG066963/AG/NIA NIH HHS/United States
- K99/R00AG049958/U.S. Department of Health & Human Services | NIH | National Institute on Aging (U.S. National Institute on Aging)
- EDUC2-12691/California Institute for Regenerative Medicine (CIRM)
- na/Arthritis National Research Foundation (ANRF)
- R01 AG070647/AG/NIA NIH HHS/United States
- na/American Federation for Aging Research (American Federation for Aging Research, Inc.)
- na/Thomas and Stacey Siebel Foundation (Siebel Foundation)
- T32 AR079099/AR/NIAMS NIH HHS/United States
- K99/R00AG066963/U.S. Department of Health & Human Services | NIH | National Institute on Aging (U.S. National Institute on Aging)
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

