Development and multi-cohort validation of a machine learning-based simplified frailty assessment tool for clinical risk prediction
- PMID: 40817251
- PMCID: PMC12357463
- DOI: 10.1186/s12967-025-06728-4
Development and multi-cohort validation of a machine learning-based simplified frailty assessment tool for clinical risk prediction
Abstract
Background: Frailty significantly impacts health outcomes in aging populations, yet its routine assessment remains challenging due to the complexity and time-consuming nature of existing tools. This study aimed to develop and validate a clinically feasible, machine learning-based frailty assessment tool that balances predictive accuracy with implementation simplicity in real-world clinical settings.
Methods: We conducted a multi-cohort study leveraging data from the National Health and Nutrition Examination Survey (NHANES, n = 3,480), China Health and Retirement Longitudinal Study (CHARLS, n = 16,792), China Health and Nutrition Survey (CHNS, n = 6,035), and Sun Yat-sen University Third Affiliated Hospital CKD cohort (SYSU3 CKD, n = 2,264). Through systematic application of five complementary feature selection algorithms to 75 potential variables, followed by comparative evaluation of 12 machine learning approaches, we developed a parsimonious assessment tool for predicting frailty diagnosis, chronic kidney disease progression, cardiovascular events, and all-cause mortality.
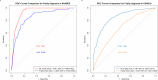
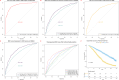
Results: Our analysis identified a minimal set of just eight readily available clinical parameters— age, sex, body mass index (BMI), pulse pressure, creatinine, hemoglobin, and preparing meals difficulty and lifting/carrying difficulty—that demonstrated robust predictive power. The extreme gradient boosting (XGBoost) algorithm exhibited superior performance across training (AUC 0.963, 95% CI: 0.951–0.975), internal validation (AUC 0.940, 95% CI: 0.924–0.956), and external validation (AUC 0.850, 95% CI: 0.832–0.868) datasets. This model significantly outperformed traditional frailty indices in predicting CKD progression (AUC 0.916 vs. 0.701, p < 0.001), cardiovascular events (AUC 0.789 vs. 0.708, p < 0.001), and mortality (time-dependent AUC 0.767 − 0.702 vs. 0.690 − 0.627, p < 0.001). SHAP analysis provided transparent insights into model predictions, facilitating clinical interpretation.
Conclusion: Our simplified frailty assessment tool demonstrates robust performance across multiple health outcomes while minimizing measurement burden. The model’s superior predictive capabilities for CKD progression, cardiovascular events, and mortality underscore its potential utility for risk stratification.
Supplementary Information: The online version contains supplementary material available at 10.1186/s12967-025-06728-4.
Keywords: Cardiovascular risk; Chronic kidney disease; Frailty assessment; Machine learning; Mortality prediction; Risk stratification; XGBoost.
Conflict of interest statement
Declarations. Ethics approval and consent to participate: This study analyzed publicly available deidentified data from NHANES 1999–2018, CHARLS, CHNS, and SYSU3 CKD cohorts. For the NHANES, CHARLS, and CHNS datasets, ethical approval was not required as these are publicly available anonymized datasets where participants had already provided informed consent for research use during the original data collection. For the SYSU3 CKD cohort, this study was approved by the Ethics Committee of the Third Affiliated Hospital of Sun Yat-Sen University (Approval No. [2023]02-358-01), and the requirement for individual informed consent was waived due to the retrospective nature of the analysis and the use of deidentified data. Consent for publication: Not applicable as no individual person’s data are contained in this manuscript in any identifiable form. Competing interests: The authors declare that they have no competing interests. None of the authors have financial or non-financial interests that may be relevant to the submitted work.
Figures



References
-
- Chan GC-K, Kalantar-Zadeh K, Ng JK-C, Tian N, Burns A, Chow K-M, et al. Frailty in patients on dialysis. Kidney Int. 2024;106:35–49. - PubMed
-
- Hoogendijk EO, Afilalo J, Ensrud KE, Kowal P, Onder G, Fried LP. Frailty: implications for clinical practice and public health. Lancet (lond Engl). 2019;394:1365–75. - PubMed
-
- Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, et al. Frailty in older adults: evidence for a phenotype. Journals Gerontol Ser A: Biol Sci Med Sci. 2001;56:M146–57. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

