Morphology and Glycan Composition of the Mandibular Glands in the White-Eared Opossum (Didelphis albiventris)
- PMID: 40817713
- PMCID: PMC12357166
- DOI: 10.1002/jmor.70074
Morphology and Glycan Composition of the Mandibular Glands in the White-Eared Opossum (Didelphis albiventris)
Abstract
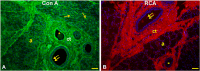
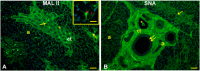
The white-eared opossum, Didelphis albiventris, is an opportunistic and omnivorous marsupial, whose diet ranges from wild fruits to eggs and birds. Salivary glycoproteins play a key role in the protection of the oral cavity and the formation of the food bolus. Despite the importance of salivary glycoproteins, their detailed investigation in the white-eared is lacking. This study investigated the morphology and glycan composition of the mandibular salivary glands of the white-eared opossum for the first time. Histological and histochemical investigations were conducted on tissue fragments fixed with 4% PBS-buffered paraformaldehyde and embedded in Paraplast. The pattern of glycoproteins was investigated using traditional histochemical methods (PAS, Alcian Blue pH 2.5, and High-Iron Diamine staining) and lectin histochemistry. The glandular parenchyma consisted of acinar secretory units and a duct system characterized by abundant striated ducts. Secretory acini secrete neutral glycans and non-sulfated acid glycans. Mannosylated N-linked glycans terminating in α2,6-sialic acid and fucose are expressed in the secretory acini, containing intraluminal α2,3-sialylated O-linked glycans. The epithelial lining of the striated and interlobular ducts also shows O-linked glycans with terminal Galβ1, 3GalNAc, and αGalNAc residues. Finally, the epithelium and lumen of interlobular ducts are enriched with additional GalNAc-terminated O-linked glycans with the appearance of lactosaminated glycans and the disappearance of α2,3-sialylated glycans. These results suggest that the saliva produced by the mandibular gland of the white-eared opossum consists of a species-specific pattern of glycoproteins, to whose composition the ductal system also contributes. The observed glycan composition is probably related to the diet of the white-eared opossum and its adaptations to the environment and food availability. These results indicate that the mandibular salivary gland of the white-eared opossum Didelphis albiventris has specific histological and molecular characteristics compared to other marsupial species, suggesting that diet and habitat, but not the taxonomic group, influence the mandibular gland features.
Keywords: glycans; lectins; marsupials; salivary glands.
© 2025 The Author(s). Journal of Morphology published by Wiley Periodicals LLC.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures





Similar articles
-
Morphology, glicohistochemistry and immunolocalization of Aquaporin 5 in the mandibular gland of the giant anteater (Myrmecophaga tridactyla).Ann Anat. 2025 Feb;258:152367. doi: 10.1016/j.aanat.2024.152367. Epub 2024 Dec 4. Ann Anat. 2025. PMID: 39643063
-
Helminths assemblage in two opossum's species, Didelphis albiventris and Didelphis aurita (Mammalia: Didelphimorphia), from the Atlantic Forest of Argentina.Parasitology. 2025 Apr;152(5):551-562. doi: 10.1017/S0031182025000563. Parasitology. 2025. PMID: 40269638 Free PMC article.
-
Urinalysis in the white-eared opossum (Didelphis albiventris).Vet Res Commun. 2025 Jul 8;49(5):248. doi: 10.1007/s11259-025-10818-5. Vet Res Commun. 2025. PMID: 40627231
-
The role of carbohydrate recognition during human sperm-egg binding.Hum Reprod. 2013 Mar;28(3):566-77. doi: 10.1093/humrep/des447. Epub 2013 Jan 12. Hum Reprod. 2013. PMID: 23315069
-
Management of urinary stones by experts in stone disease (ESD 2025).Arch Ital Urol Androl. 2025 Jun 30;97(2):14085. doi: 10.4081/aiua.2025.14085. Epub 2025 Jun 30. Arch Ital Urol Androl. 2025. PMID: 40583613 Review.
References
-
- Accogli, G. , Crovace A. M., Mastrodonato M., Rossi G., Francioso E. G., and Desantis S.. 2018. “Probiotic Supplementation Affects the Glycan Composition of Mucins Secreted by Brunner's Glands of the Pig Duodenum.” Annals of Anatomy ‐ Anatomischer Anzeiger 218: 236–242. 10.1016/j.aanat.2018.03.008. - DOI - PubMed
-
- Adnyane, I. K. M. , Zuki A. B., Noordin M. M., and Agungpriyono S.. 2010. “Histological Study of the Parotid and Mandibular Glands of Barking Deer (Muntiacus muntjak) With Special Reference to the Distribution of Carbohydrate Content.” Anatomia, Histologia, Embryologia 39: 516–520. 10.1111/j.1439-0264.2010.01023.x. - DOI - PubMed
-
- Bitencourt, M. M. , and Bezerra A. M. R.. 2022. “Infection Agents of Didelphidae (Didelphimorphia) of Brazil: An Underestimated Matter in Zoonoses Research.” Mammalia 86: 105–122. 10.1515/mammalia-2021-0134. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

