Hematological parameters as predictors of immune-related adverse events: risk factor analysis in non-small cell lung cancer patients undergoing immunotherapy
- PMID: 40821056
- PMCID: PMC12351628
- DOI: 10.62347/EUHL7337
Hematological parameters as predictors of immune-related adverse events: risk factor analysis in non-small cell lung cancer patients undergoing immunotherapy
Abstract
Objective: To evaluate the predictive value of hematological biomarkers in assessing the risk of immune-related adverse events (irAEs) in non-small cell lung cancer (NSCLC) patients undergoing immunotherapy and to identify potential risk factors for personalized treatment optimization.
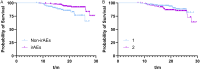
Methods: Clinical data of 274 NSCLC patients who received immunotherapy between April 2018 and January 2021 were retrospectively analyzed. Patients were divided into irAEs and non-irAEs groups based on the occurrence of irAEs. Peripheral blood indices within one week before treatment initiation were assessed and compared, including absolute neutrophil count (ANC), lymphocyte count (LYM), neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), systemic immune-inflammation index (SII), albumin-to-alkaline phosphatase ratio (AAPR), and albumin-to-fibrinogen ratio (AFR). Kaplan-Meier analysis compared overall survival (OS) and progression-free survival (PFS), while logistic regression identified independent risk factors for irAEs. Receiver operating characteristic (ROC) curve analysis evaluated predictive performance.
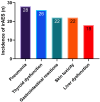
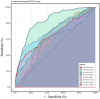
Results: Among the 274 patients, 116 (42.2%) developed irAEs. Compared to the non-irAEs group, the irAEs group exhibited significantly higher ANC, NLR, PLR, and SII, along with lower LYM, AAPR, and AFR as well as lower OS and PFS rates (all P < 0.05). Logistic regression showed that all hematological indicators were independent risk factors for irAEs (P < 0.05). ROC analysis showed an AUC of 0.722 for NLR and 0.829 for the combined model.
Conclusion: Pretreatment assessment of ANC, LYM, NLR, PLR, SII, AAPR, and AFR provides valuable predictive utility for irAEs risk in NSCLC patients undergoing immunotherapy. Integrating these biomarkers into clinical practice may enhance risk stratification and guide personalized treatment strategies to improve safety and therapeutic outcomes.
Keywords: Hematological indicators; immune-related adverse events; immunotherapy; non-small cell lung cancer; risk factors.
AJTR Copyright © 2025.
Conflict of interest statement
None.
Figures

Similar articles
-
Predictive value of systemic inflammatory indices for perinatal outcomes following cervical cerclage: a retrospective cohort study.BMC Pregnancy Childbirth. 2025 Jul 10;25(1):750. doi: 10.1186/s12884-025-07888-3. BMC Pregnancy Childbirth. 2025. PMID: 40640782 Free PMC article.
-
Predictive value of NLR, PLR and MPVLR for recent major cardiovascular adverse events in elderly patients with heart failure.Am J Transl Res. 2025 Jul 15;17(7):4964-4975. doi: 10.62347/XQNT2355. eCollection 2025. Am J Transl Res. 2025. PMID: 40821098 Free PMC article.
-
Predictive value of traditional laboratory parameters and inflammatory indices for same-day seizure recurrence in patients with epileptic seizures.Epilepsia Open. 2025 Aug 18. doi: 10.1002/epi4.70123. Online ahead of print. Epilepsia Open. 2025. PMID: 40824701
-
Systemic treatments for metastatic cutaneous melanoma.Cochrane Database Syst Rev. 2018 Feb 6;2(2):CD011123. doi: 10.1002/14651858.CD011123.pub2. Cochrane Database Syst Rev. 2018. PMID: 29405038 Free PMC article.
-
Comparison of efficacy and safety of PD-1/PD-L1 combination therapy in first-line treatment of advanced NSCLC: an updated systematic review and network meta-analysis.Clin Transl Oncol. 2024 Oct;26(10):2488-2502. doi: 10.1007/s12094-024-03442-3. Epub 2024 Apr 16. Clin Transl Oncol. 2024. PMID: 38625495
References
-
- Shankar B, Zhang J, Naqash AR, Forde PM, Feliciano JL, Marrone KA, Ettinger DS, Hann CL, Brahmer JR, Ricciuti B, Owen D, Toi Y, Walker P, Otterson GA, Patel SH, Sugawara S, Naidoo J. Multisystem immune-related adverse events associated with immune checkpoint inhibitors for treatment of non-small cell lung cancer. JAMA Oncol. 2020;6:1952–1956. - PMC - PubMed
-
- Nuñez NG, Berner F, Friebel E, Unger S, Wyss N, Gomez JM, Purde MT, Niederer R, Porsch M, Lichtensteiger C, Kramer R, Erdmann M, Schmitt C, Heinzerling L, Abdou MT, Karbach J, Schadendorf D, Zimmer L, Ugurel S, Klümper N, Hölzel M, Power L, Kreutmair S, Capone M, Madonna G, Cevhertas L, Heider A, Amaral T, Hasan Ali O, Bomze D, Dimitriou F, Diem S, Ascierto PA, Dummer R, Jäger E, Driessen C, Levesque MP, van de Veen W, Joerger M, Früh M, Becher B, Flatz L. Immune signatures predict development of autoimmune toxicity in patients with cancer treated with immune checkpoint inhibitors. Med. 2023;4:113–129. e117. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous
