Redox-Sensitive Camptothecin Prodrug: A Promising Drug Delivery Strategy with Ultrahigh Drug Loading and Tunable Drug Release
- PMID: 40821452
- PMCID: PMC12355937
- DOI: 10.1016/j.ntm.2025.100088
Redox-Sensitive Camptothecin Prodrug: A Promising Drug Delivery Strategy with Ultrahigh Drug Loading and Tunable Drug Release
Abstract
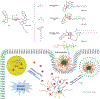
Small molecular drugs play a critical role in cancer therapy but face challenges like poor solubility, severe side effects, and inefficient delivery. Polymeric micellar-based drug delivery systems show promise but struggle with low drug loading, instability, and premature drug release partly due to the incompatible physicochemical properties. Here, we report a simple and efficient method to develop redox-sensitive camptothecin (CPT) prodrug by conjugating alkyl chains to CPT via a disulfide linker. By conjugating alkyl chains of varying lengths to CPT via a disulfide linker, we achieved high drug-loading efficiency and loading capacity, controlled responsive drug release, due to enhanced hydrophobic interaction and miscibility with the carrier. The prodrug loaded NPs exhibited slower drug release for more hydrophobic ones with longer alkyl chains. In vitro cytotoxicity assays against cancer cells confirmed the prodrugs' potency and the critical role of the disulfide bond in maintaining anticancer activity. These findings highlight the importance of tuning prodrug hydrophobicity and GSH sensitivity in drug delivery. This prodrug engineering strategy, which involves conjugating a hydrophobic alkyl chain to modulate the drug's physicochemical properties, offers a straightforward approach for designing and optimizing drug delivery systems for a wide range of therapeutic agents, whether hydrophilic or hydrophobic.
Keywords: Camptothecin; Chemotherapeutic agent; Drug delivery; Nanomedicine; Ultrahigh drug loading.
Figures


Similar articles
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
Balancing Chemical and Supramolecular Stability in OEGylated Supramolecular Polymers for Systemic Drug Delivery.J Am Chem Soc. 2025 May 28;147(21):17985-17993. doi: 10.1021/jacs.5c03253. Epub 2025 May 15. J Am Chem Soc. 2025. PMID: 40375654 Free PMC article.
-
Structure-guided dual modification prodrug strategy for optimizing camptothecin delivery in pancreatic cancer chemotherapy.Int J Pharm. 2025 Sep 15;682:125908. doi: 10.1016/j.ijpharm.2025.125908. Epub 2025 Jun 27. Int J Pharm. 2025. PMID: 40582523
-
Chemotherapy for advanced gastric cancer.Cochrane Database Syst Rev. 2017 Aug 29;8(8):CD004064. doi: 10.1002/14651858.CD004064.pub4. Cochrane Database Syst Rev. 2017. PMID: 28850174 Free PMC article.
-
Mechanistic insight into nanomedicine for polycystic ovary syndrome.Mol Biol Rep. 2025 Jun 21;52(1):618. doi: 10.1007/s11033-025-10709-7. Mol Biol Rep. 2025. PMID: 40544202 Review.
References
-
- Wang X, Zhuang Y, Wang Y, Jiang M, Yao L, The recent developments of camptothecin and its derivatives as potential anti-tumor agents, European Journal of Medicinal Chemistry 260 (2023) 115710. - PubMed
-
- Krishnan P, Rajan M, Kumari S, Sakinah S, Priya SP, Amira F, Danjuma L, Pooi Ling M, Fakurazi S, Arulselvan P, Higuchi A, Arumugam R, Alarfaj AA, Munusamy MA, Hamat RA, Benelli G, Murugan K, Kumar SS, Efficiency of newly formulated camptothecin with β-cyclodextrin-EDTA-Fe3O4 nanoparticle-conjugated nanocarriers as an anti-colon cancer (HT29) drug, Scientific Reports 7(1) (2017) 10962. - PMC - PubMed
-
- Martino E, Della Volpe S, Terribile E, Benetti E, Sakaj M, Centamore A, Sala A, Collina S, The long story of camptothecin: From traditional medicine to drugs, Bioorganic & Medicinal Chemistry Letters 27(4) (2017) 701–707. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
