Development of a highly efficient p-coumaric acid-responsive biosensor in Saccharomyces cerevisiae
- PMID: 40821659
- PMCID: PMC12355551
- DOI: 10.1016/j.synbio.2025.07.007
Development of a highly efficient p-coumaric acid-responsive biosensor in Saccharomyces cerevisiae
Abstract
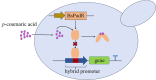
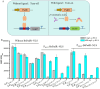
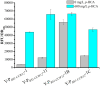
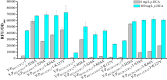
Developing biosensors to monitor and regulate intracellular biosynthesis pathways can significantly enhance natural product yields in microbial cell factories. This study created a novel biosensor in Saccharomyces cerevisiae to respond to p-coumaric acid, a critical precursor in the biosynthesis of polyphenols and flavonoids. This biosensor was constructed by expressing the BsPadR repressor from Bacillus subtilis and engineering hybrid promoters. Notably, the P BS1-CCW12 hybrid promoter exhibited tight regulation by BsPadR and enhanced activity in response to p-coumaric acid. However, excessive BsPadR expression negatively impacted yeast growth, which was mitigated by using weaker promoters, P BST1 and P ERG9 . Furthermore, the impact of nuclear localization signal (SV40-NLS) positioning on BsPadR functionality was explored, revealing that fusion of an SV40-NLS at the C-terminus of BsPadR enhanced the biosensor's performance. To validate its utility, we applied this system to dynamically regulate CrtE (geranylgeranyl pyrophosphate synthase), a key enzyme in lycopene biosynthesis. By coupling p-coumaric acid production with lycopene biosynthesis, we enabled high-throughput colorimetric screening for enzyme evolution and strain selection. This novel biosensor serves as a valuable tool for future studies aimed at optimizing the production of p-coumaric acid and its derivatives in S. cerevisiae, thereby advancing the efficiency of biosynthetic processes in microbial cell factories.
Keywords: Biosensor; BsPadR transcriptional repressor; Hybrid promoter; Saccharomyces cerevisiae; p-Coumaric acid.
© 2025 The Authors.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures



Similar articles
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
De Novo Synthesis of Friedelin in Saccharomyces cerevisiae via Combination of Metabolic and Lipid Droplet Engineering.ACS Synth Biol. 2025 Jun 20;14(6):2130-2138. doi: 10.1021/acssynbio.5c00047. Epub 2025 May 15. ACS Synth Biol. 2025. PMID: 40373267
-
High-Throughput Indirect Monitoring of TORC1 Activation Using the pTOMAN-G Plasmid in Yeast.Bio Protoc. 2025 Jun 20;15(12):e5356. doi: 10.21769/BioProtoc.5356. eCollection 2025 Jun 20. Bio Protoc. 2025. PMID: 40620813 Free PMC article.
-
Isolated Methylmalonic Acidemia.2005 Aug 16 [updated 2022 Sep 8]. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2025. 2005 Aug 16 [updated 2022 Sep 8]. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2025. PMID: 20301409 Free Books & Documents. Review.
-
Management of urinary stones by experts in stone disease (ESD 2025).Arch Ital Urol Androl. 2025 Jun 30;97(2):14085. doi: 10.4081/aiua.2025.14085. Epub 2025 Jun 30. Arch Ital Urol Androl. 2025. PMID: 40583613 Review.
References
-
- Otto M., Liu D., Siewers V. In: Engineering natural product biosynthesis: methods and protocols. Skellam E., editor. Springer US; 2022. Saccharomyces cerevisiae as a heterologous host for natural products; pp. 333–367. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

