Cholinotrophic basal forebrain connectome dysfunction in Down syndrome with and without dementia
- PMID: 40822354
- PMCID: PMC12357115
- DOI: 10.1016/j.isci.2025.113041
Cholinotrophic basal forebrain connectome dysfunction in Down syndrome with and without dementia
Abstract
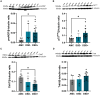
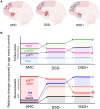
Cholinotrophic basal forebrain (CTBF) neurons depend upon the complex interaction of both upstream and downstream nerve growth factor (NGF) signaling pathways for survival and function. Although dysfunction of the NGF system occurs in both Down syndrome (DS), not all individuals with DS develop dementia. Whether NGF system dysregulation differs between demented individuals with DS (DSD+) versus those without dementia (DSD-) is unknown. Here, we report a significant reduction in neurons positive for the p75NTR within the nucleus basalis of Meynert in DSD+, but not DSD-, compared to aged-matched controls (AMC). ChAT positive cells were significantly lower in DSD+ compared to DSD- and AMC cases. FC levels of p75NTR and proNGF were increased, while ChAT levels decreased in DSD+ compared to AMC. A greater number of AT8 tau positive neuropil threads and Thioflavin-S labeled neurofibrillary tangles were found in DSD+. These findings suggest a greater role for p75NTR/proNGF in demented in individuals with DS compared to those with dementia. The factors that underlie cognitive resilience in DSD- remains to be determined.
Keywords: Cell biology; Neuroscience.
© 2025 The Authors.
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
U1-70K and U1A and tau pathogenesis in demented and non-demented individuals with Down syndrome.Alzheimers Dement. 2025 Aug;21(8):e70474. doi: 10.1002/alz.70474. Alzheimers Dement. 2025. PMID: 40762109 Free PMC article.
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
Tonsillectomy or adenotonsillectomy versus non-surgical management for obstructive sleep-disordered breathing in children.Cochrane Database Syst Rev. 2015 Oct 14;2015(10):CD011165. doi: 10.1002/14651858.CD011165.pub2. Cochrane Database Syst Rev. 2015. PMID: 26465274 Free PMC article.
-
Expression of NGF, proNGF, p75NTR in lung injury induced by cerebral ischemia-reperfusion in young and elderly rats.Clinics (Sao Paulo). 2024 Nov 15;79:100532. doi: 10.1016/j.clinsp.2024.100532. eCollection 2024. Clinics (Sao Paulo). 2024. PMID: 39549390 Free PMC article.
-
Withdrawal versus continuation of chronic antipsychotic drugs for behavioural and psychological symptoms in older people with dementia.Cochrane Database Syst Rev. 2013 Mar 28;(3):CD007726. doi: 10.1002/14651858.CD007726.pub2. Cochrane Database Syst Rev. 2013. PMID: 23543555
References
LinkOut - more resources
Full Text Sources
Research Materials

