A sprayable exosome-loaded hydrogel with controlled release and multifunctional synergistic effects for diabetic wound healing
- PMID: 40822934
- PMCID: PMC12355498
- DOI: 10.1016/j.mtbio.2025.102159
A sprayable exosome-loaded hydrogel with controlled release and multifunctional synergistic effects for diabetic wound healing
Abstract
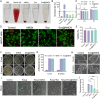
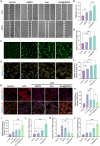
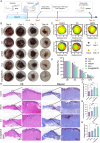
Diabetic wound healing is hindered by bacterial infections, oxidative stress, impaired vascularization, and chronic inflammation. Conventional dressings, limited by static drug release and single-functionality, fail to dynamically match the varying demands of different healing stages and dressing replacement frequencies. This study developed a multifunctional sprayable hydrogel dressing (Exo@AMCN) by photocrosslinking methacrylated decellularized dermal matrix co-loaded with human umbilical cord mesenchymal stem cell-derived exosomes (hUCMSC-Exo) and β-cyclodextrin-borneol inclusion complexes (CN). The hydrogel can be sprayed onto irregularly shaped wounds, with its crosslinking density and degradation kinetics precisely modulated by adjusting the photocuring duration. This tunability enables controlled release of exosomes and borneol over 2-7 days. Experimental findings demonstrate that Exo@AMCN displays excellent biocompatibility, broad-spectrum antibacterial activity (>85 % efficacy), and robust reactive oxygen species scavenging capacity. The dressing significantly boosts cell migration, fosters angiogenesis, and prompts macrophage polarization toward anti-inflammatory phenotypes. In a diabetic wound model, Exo@AMCN reduced residual wound area to 1.07 ± 1.27 % within 14 days by modulating tissue inflammation, enhancing collagen deposition, and stimulating neovascularization. This innovative approach, combining controlled drug release with multifunctional synergy, offers a promising individualized solution for managing diabetic wounds.
Keywords: Acellular dermal matrix; Borneol; Diabetic wound healing; Exosomes; Hydrogel.
© 2025 The Authors.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures






Similar articles
-
Spatiotemporal self-reinforcing hydrogel spray with antibacterial and anti-inflammatory properties for accelerated diabetic wound healing.Acta Biomater. 2025 Aug;202:193-204. doi: 10.1016/j.actbio.2025.06.036. Epub 2025 Jun 20. Acta Biomater. 2025. PMID: 40544984
-
A hybrid hydrogel constructed using drug loaded mesoporous silica and multiple response copolymer as an intelligent dressing for wound healing of diabetic foot ulcers.J Mater Chem B. 2023 Jun 7;11(22):4922-4933. doi: 10.1039/d3tb00395g. J Mater Chem B. 2023. PMID: 37194321
-
Block cationic copolymer/quaternary ammonium chitosan-based composite antibacterial hydrogel dressings with NIR photothermal effects for bacteria-infected wound healing.Int J Biol Macromol. 2025 Feb;288:138716. doi: 10.1016/j.ijbiomac.2024.138716. Epub 2024 Dec 12. Int J Biol Macromol. 2025. PMID: 39672433
-
The Promising Hydrogel Candidates for Preclinically Treating Diabetic Foot Ulcer: A Systematic Review and Meta-Analysis.Adv Wound Care (New Rochelle). 2023 Jan;12(1):28-37. doi: 10.1089/wound.2021.0162. Epub 2022 Apr 11. Adv Wound Care (New Rochelle). 2023. PMID: 35229628
-
Dressings and topical agents for the management of open wounds after surgical treatment for sacrococcygeal pilonidal sinus.Cochrane Database Syst Rev. 2022 May 20;5(5):CD013439. doi: 10.1002/14651858.CD013439.pub2. Cochrane Database Syst Rev. 2022. PMID: 35593897 Free PMC article.
References
-
- GBD 2021 Japan Collaborators Three decades of population health changes in Japan, 1990-2021: a subnational analysis for the global burden of disease study 2021. Lancet Public Health. 2025 Apr;10(4):e321–e332. doi: 10.1016/S2468-2667(25)00044-1. Epub 2025 Mar 20. PMID: 40122087; PMCID: PMC11959113. - DOI - PMC - PubMed
-
- Ebaid H., Ahmed O.M., Mahmoud A.M., Ahmed R.R. Limiting prolonged inflammation during proliferation and remodeling phases of wound healing in streptozotocin-induced diabetic rats supplemented with camel undenatured whey protein. BMC Immunol. 2013 Jul 25;14:31. doi: 10.1186/1471-2172-14-31. PMID: 23883360; PMCID: PMC3729579. - DOI - PMC - PubMed
-
- Jennaro T.S., Puskarich M.A., Evans C.R., Karnovsky A., Flott T.L., McLellan L.A., Jones A.E., Stringer K.A. Sustained perturbation of metabolism and metabolic subphenotypes are associated with mortality and protein markers of the host response. Crit Care Explor. 2023 Mar 27;5(4) doi: 10.1097/CCE.0000000000000881. PMID: 36998529; PMCID: PMC10047616. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

