Non-inferiority of sleep position therapy compared to positive airway pressure therapy with regard to daytime sleepiness in patients with mild to moderate position-dependent obstructive sleep apnoea (POSA): study protocol for a multicentre randomised cross-over trial
- PMID: 40826110
- PMCID: PMC12363012
- DOI: 10.1186/s13063-025-09007-1
Non-inferiority of sleep position therapy compared to positive airway pressure therapy with regard to daytime sleepiness in patients with mild to moderate position-dependent obstructive sleep apnoea (POSA): study protocol for a multicentre randomised cross-over trial
Abstract
Background: First-line therapy for patients with clinically relevant obstructive sleep apnoea (OSA) is positive airway pressure therapy (PAP). At least one half of patients with mild to moderate OSA (apnoea-hypopnoea-index (AHI) 5-30/h) have positional OSA (POSA), where apnoea occurs mostly in supine sleep. These patients might benefit from sleep-position therapy (SPT) which should reduce sleeping time spent in a supine position. Until now, it is unclear whether SPT is non-inferior to PAP therapy for symptom relief in these patients.
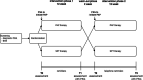
Methods: This is a multicentre, non-inferiority, open-label randomised, cross-over clinical trial. Patients with mild to moderate POSA and daytime sleepiness (according to Epworth Sleepiness Scale (ESS) > 10 points) will be randomised with a 1:1 allocation ratio, stratified by centre and AHI, to start with either PAP therapy or SPT and treated for 12 weeks. After a wash-out period of two weeks, they will switch to the other therapy for 12 weeks. The primary outcome is daytime sleepiness measured by the ESS at the end of each treatment phase. Analysis will be done in the intention-to-treat population using a linear mixed-effects model containing the intervention, the phase, the interaction between therapy and phase (including the carry-over effect) and the baseline measurement of the ESS and AHI as fixed effect, and centre and patient as random effects. A one-sided test at significant level of 2.5% will be used to test the non-inferiority of SPT with a non-inferiority margin of 1.35. Based on a sample size calculation with a one-sided one-sample t-test at significant level of 2.5%, assuming a standard deviation of 4, a total of 418 patients should be included to reach 80% power when SPT is only slightly inferior to PAP therapy (difference 0.8 resulting in a delta of 0.55 (1.35-0.8)). Assuming a 5% drop-out rate, 220 patients per sequence should be included. Possible futility stopping is planned at an interim analysis after 300 patients.
Discussion: The recruitment of patients with mild to moderate POSA is feasible with the planned centres. Both certified interventions (PAP and SPT) are covered by the statutory health insurance companies as part of the trial guideline.
Trial registration: DRKS00033048 registered 17. June 2024, http://www.drks.de .
Keywords: Daytime sleepiness; Mild to moderate position-dependent obstructive sleep apnoea; Positive airway pressure therapy; Sleep position therapy.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate {24}: Ethical approval was obtained on 22 May 2024. All participating patients will provide written informed consent. Consent for publication {32}: Not applicable. Competing interests {28}: NT, FV, AS, JW, AN: The authors declare that they have no competing interests. CS received institutional lecture and consulting fees from Astra-Zeneca, Berlin-Chemie, Bioprojet, Bristol-Myers Squibb, Chiesi, Eli Lilly, Idorsia, Inspire, Jazz, Mementor, Philips-Respironics, Pfizer, ResMed, ZOLL. He also received institutional research grants from Bayer, Mementor, Nox Medical, ResMed, Sleepiz as well as public funding by the German Federal Ministry for Economic Affairs and Climate Action, German Federal Ministry of Education and Research, German Federal Joint Committee, German Research Foundation.
References
-
- Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep. 1991;14(6):540–5. Available from: https://academic.oup.com/sleep/article/14/6/540/2742871?login=true. - PubMed
-
- Lévy P, Kohler M, McNicholas WT, Barbé F, McEvoy RD, Somers VK, et al. Obstructive sleep apnoea syndrome. Nat Rev Dis Primers. 2015;1:15015. - PubMed
-
- Richtlinie des Gemeinsamen Bundesausschusses zu Untersuchungs- und Behandlungsmethoden der vertragsärztlichen Versorgung. Available from: URL: https://www.g-ba.de/downloads/62-492-2901/MVV-RL-2022-05-19-iK-2022-08-0.... Cited 2022 Nov 2.
-
- Deutsche Gesellschaft für Schlafforschung. S3-Leitlinie Nicht erholsamer Schlaf/Schlafstörungen – Kapitel „Schlafbezogene Atmungsstörungen“: S3-Leitlinie Schlafbezogene Atmungsstörungen. Available from: URL: https://www.awmf.org/leitlinien/detail/ll/063-001.html. Cited 2022 Nov 2.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials