Real-World Prevalence and Outcomes of Patients with Paroxysmal Nocturnal Hemoglobinuria Treated with C5 Inhibitors in the US: A Retrospective Claims Database Analysis
- PMID: 40827257
- PMCID: PMC12358179
- DOI: 10.36469/001c.142049
Real-World Prevalence and Outcomes of Patients with Paroxysmal Nocturnal Hemoglobinuria Treated with C5 Inhibitors in the US: A Retrospective Claims Database Analysis
Abstract
Background: Paroxysmal nocturnal hemoglobinuria (PNH) is a rare blood disorder with C5 inhibitors (C5i), eculizumab and ravulizumab, being part of current treatment options.
Objectives: To estimate the 5-year prevalence of PNH and describe the healthcare resource utilization and direct healthcare costs associated with C5i among commercially insured patients with PNH treated with C5i in the US.
Methods: The 5-year prevalence of adults with PNH in IQVIA PharMetrics® Plus was estimated (2018-2022). A retrospective cohort study (2011-2022) was also conducted in adults with PNH treated with C5i and ≥3 months of continuous health plan coverage following the first claim for C5i (index date). PNH-related health resource utilization and direct healthcare costs were assessed from index date until earliest of treatment discontinuation/end of data/end of continuous health plan coverage (follow-up period).
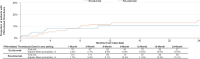
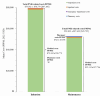
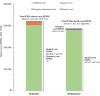
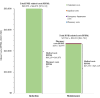
Results: The 5-year prevalence of PNH was 2.4 per 100 000 persons in commercial claims. A total of 371 patients treated with C5i (median age: 40 years; female: 55.3%; eculizumab: 53.9%; ravulizumab: 46.1%) were followed for a mean ± SD [median] of 19.3 ± 16.9 [14.7] months. Annual incidence rates of PNH-related blood transfusion and breakthrough hemolysis (BTH) among patients treated with C5i were 1.2 (eculizumab: 1.3; ravulizumab: 1.0) and 4.5 (eculizumab: 5.2; ravulizumab: 3.3) per person per year (PPPY), respectively. In patients treated with eculizumab and ravulizumab, respectively, PNH-related blood transfusion was required by 46.2% and 11.9% of patients in the first 6 months post-index, and over the follow-up period, transfusion avoidance was observed in 46.2% and 78.2% of patients. The 6- and 12-month rates of PNH-related thrombosis were 8.0% and 10.6% for eculizumab and 6.1% and 11.6% for ravulizumab, respectively. Among patients treated with C5i, estimated annual total PNH-related costs PPPY were 697 459; ravulizumab: 633 984 (eculizumab: 570 832) for subsequent years, with treatment costs accounting for 94.3% to 94.6% of total costs.
Discussion: Despite treatment with C5i, patients with PNH still exhibited BTH, required blood transfusions, and experienced thrombosis.
Conclusion: This study highlights the unmet need for more effective PNH treatments to address the economic and clinical burden associated with PNH and improve disease control among patients.
Keywords: blood transfusion; breakthrough hemolysis; direct healthcare costs; eculizumab; healthcare resource utilization; paroxysmal nocturnal hemoglobinuria; ravulizumab; thrombosis.
Conflict of interest statement
S.T. has provided paid consulting services to Novartis Pharmaceuticals Corporation, which funded the development and conduct of this study and manuscript. S.L., J.P., L.G., and G.Y. are employees of Novartis Pharmaceuticals Corporation. M.C. was an employee of Novartis Pharmaceutical Corporation at the time that this study was conducted. D.L.-V., R.D., S.S. and A.G. are employees of Analysis Group, Inc., a consulting company that has provided paid consulting services to Novartis Pharmaceuticals Corporation, which funded the development and conduct of this study and manuscript.
Figures
Similar articles
-
Ravulizumab demonstrates long-term efficacy, safety and favorable patient survival in patients with paroxysmal nocturnal hemoglobinuria.Ann Hematol. 2025 Jan;104(1):81-94. doi: 10.1007/s00277-025-06193-5. Epub 2025 Jan 22. Ann Hematol. 2025. PMID: 39841198 Free PMC article. Clinical Trial.
-
Long-term effectiveness and safety of ravulizumab in paroxysmal nocturnal hemoglobinuria: a plain language summary.J Comp Eff Res. 2025 Aug;14(8):e250022. doi: 10.57264/cer-2025-0022. Epub 2025 Jul 4. J Comp Eff Res. 2025. PMID: 40611794 Free PMC article. Review.
-
Descriptive, real-world treatment patterns, resource use, and total cost of care among eculizumab- and ravulizumab-treated members with paroxysmal nocturnal hemoglobinuria.J Manag Care Spec Pharm. 2023 Aug;29(8):941-951. doi: 10.18553/jmcp.2023.29.8.941. J Manag Care Spec Pharm. 2023. PMID: 37523317 Free PMC article.
-
Eculizumab for treating patients with paroxysmal nocturnal hemoglobinuria.Cochrane Database Syst Rev. 2014 Oct 30;(10):CD010340. doi: 10.1002/14651858.CD010340.pub2. Cochrane Database Syst Rev. 2014. PMID: 25356860
-
Changes in Hematologic Lab Measures Observed in Patients with Paroxysmal Nocturnal Hemoglobinuria Treated with C5 Inhibitors, Ravulizumab and Eculizumab: Real-World Evidence from a US Based EMR Network.Hematol Rep. 2023 Apr 21;15(2):266-282. doi: 10.3390/hematolrep15020027. Hematol Rep. 2023. PMID: 37092521 Free PMC article.
LinkOut - more resources
Full Text Sources
Miscellaneous
