A competing risk analysis of predictors of time to lost to follow-up among adults with TB/HIV coinfection in Bahir Dar
- PMID: 40830417
- PMCID: PMC12365281
- DOI: 10.1038/s41598-025-15985-8
A competing risk analysis of predictors of time to lost to follow-up among adults with TB/HIV coinfection in Bahir Dar
Abstract
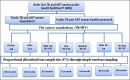
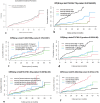
Lost to follow-up (LTFU), defined as interrupting anti-TB treatment for ≥ 8 consecutive weeks or missing anti-retroviral therapy (ART) appointments for > 90 days, is a barrier to TB/HIV coinfection management. Poor treatment adherence, a driver of multidrug resistance in TB/HIV, poses critical challenges to case management. Overestimating effect sizes when considering mutually exclusive events, like LTFU from ART and anti-TB treatment, can occur if sources of error are not properly accounted for in competing events. However, studies estimating the effect sizes of predictors of time to LTFU using competing risk analysis are scarce. Hence, this study aimed to investigate the predictors of time to LTFU among adults with TB/HIV coinfection. We conducted a multicenter facility-based retrospective follow-up study. We randomly selected 471 TB/HIV coinfected adults. Data were extracted using standardised checklists. The LTFUs from ART and anti-TB treatment were events of interest and competing events, respectively, and others were censored. The data were entered into Epi data and then exported to Stata and Rstudio. Statistical differences were tested by Gray's test, and the cumulative incidence of each event was estimated by a cumulative incidence function. Bivariable and multivariable competing risk regression models were fitted, and variables with p values < 0.05 were considered significant predictors. Incidence rates of LTFU for ART and TB treatment were 3.90 and 19.17 per 1000 person-months of observation (PMOs), respectively. The predictors of ART LTFU included rural residence (adjusted subdistribution hazard ratio (SDHR): 3.39), WHO stage IV (SDHR: 2.88), haemoglobin < 11 g/dl (SDHR: 3.56), and opportunistic infections (OIs) (SDHR: 3.65). For TB treatment LTFU, significant predictors were rural residence (SDHR: 0.11), divorced (SDHR: 2.81), widowed (SDHR: 5.92), BMI < 18.5 (SDHR: 0.41), ambulatory functional status (SDHR: 2.59), adverse drug effects (SDHR: 2.87), and poor ART adherence (SDHR: 5.72). Considering errors in competing events, ART LTFU was higher among rural dwellers, individuals with advanced disease, nutritional deficits, or adverse drug effects requiring prioritised, multifaceted interventions. Targeted strategies such as intensified monitoring, adherence counselling, nutritional support, proactive management of drug-related side effects, marital instability, OIs and poor ART adherence should be integrated into the existing ART/TB program to mitigate LTFU.
Keywords: Competing risk regression analysis; TB/HIV coinfection; Time to LTFU.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests. Ethical approval and consent to participate: Due to the retrospective nature of the study, the need to obtain the informed consent was waived by the Institutional Review Board (IRB) of the College of Health Sciences of Bahir Dar University approved the study, with study protocol number 730/2023. The IRB performs following the Declaration of Helsinki, the International Conferences on Harmonisation (ICH) Good Clinical Practice, the WHO Operating Guidelines for Ethical Review Committee and the National Guideline for Research Ethics in Ethiopia. The Amhara Public Health Institute (APHI) reviewed the ethical approval protocols and requested that the selected hospitals and health centres provide records of TB/HIV treatment follow-up data to the authors. Accordingly, the health facilities’ patient charts or ART follow-up forms were used. The name of the patient was not included in the checklist. The data of any patient were not used for other purposes other than for the aim of this study.
Figures
Similar articles
-
Retrospective cohort study of predictors of loss to follow up among adolescents and young adults living with HIV on ART in Dar es Salaam, Tanzania, 2015-2019.BMC Infect Dis. 2025 Jul 1;25(1):852. doi: 10.1186/s12879-025-11157-1. BMC Infect Dis. 2025. PMID: 40597756 Free PMC article.
-
Time-dependent predictors of loss to follow-up in HIV care in low-resource settings: A competing risks approach.PLoS One. 2025 Jul 24;20(7):e0329132. doi: 10.1371/journal.pone.0329132. eCollection 2025. PLoS One. 2025. PMID: 40705795 Free PMC article.
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
The Effect of Early Initiation of Antiretroviral Therapy in TB/HIV-Coinfected Patients: A Systematic Review and Meta-Analysis.J Int Assoc Provid AIDS Care. 2015 Nov-Dec;14(6):560-70. doi: 10.1177/2325957415599210. Epub 2015 Aug 19. J Int Assoc Provid AIDS Care. 2015. PMID: 26289343
-
Optimisation of antiretroviral therapy in HIV-infected children under 3 years of age.Cochrane Database Syst Rev. 2014 May 22;2014(5):CD004772. doi: 10.1002/14651858.CD004772.pub4. Cochrane Database Syst Rev. 2014. PMID: 24852077 Free PMC article.
References
-
- Lawn, S. D. & Zumla, A. I. Tuberculosis. Lancet378 (9785), 57–72. (2011). - PubMed
-
- WHO G. Global tuberculosis report 2020. Glob. Tuberc. Rep. (2020).
-
- Organization, W. H. Global Tuberculosis Report 2021: Supplementary Material (World Health Organization, 2022).
-
- McIlleron, H., Meintjes, G., Burman, W. J. & Maartens, G. Complications of antiretroviral therapy in patients with tuberculosis: drug interactions, toxicity, and immune reconstitution inflammatory syndrome. J. Infect. Dis.196 (1), S63–75 (2007). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical